Comparative analysis of approaches in renal cell carcinoma pharmacotherapy in different countries and in Ukraine
DOI:
https://doi.org/10.15587/2519-4852.2026.353172Keywords:
renal cell carcinoma, pharmacotherapy, immunotherapy, targeted therapy, clinical guidelines, regulatory frameworkAbstract
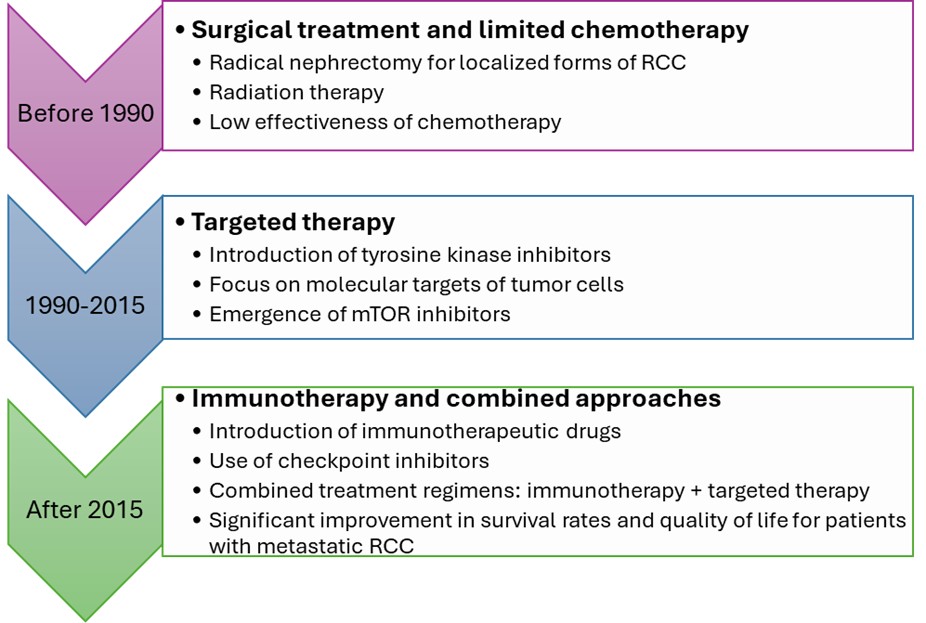
Renal cell carcinoma (RCC) is one of the most common malignant kidney tumors, characterized by a steady increase in incidence and high mortality worldwide, including in Ukraine, which necessitates timely diagnosis and comprehensive multimodal treatment.
The aim of the study was to examine approaches to RCC pharmacotherapy in different countries to further develop measures to improve pharmaceutical care for RCC patients in Ukraine.
Materials and methods. The study was based on scientific publications on RCC pharmacotherapy, international RCC treatment guidelines - National Comprehensive Cancer Network (USA), European Society for Medical Oncology (EU), and American Society of Clinical Oncology (USA) - national RCC treatment protocols, as well as Ukrainian regulatory legal acts governing pharmaceutical care for oncology patients. Content analysis, comparison, information synthesis, and analytical review were used.
Results. International guidelines emphasize personalized therapy, wide use of immunotherapy–targeted therapy combinations, IMDC-based risk stratification, and multidisciplinary patient management. Combinations such as pembrolizumab + axitinib, nivolumab + cabozantinib, and nivolumab + ipilimumab are recommended as first-line therapy for metastatic RCC, while adjuvant pembrolizumab is indicated for patients at high risk of recurrence. The Ukrainian clinical protocol partially aligns with these standards but remains limited in immunotherapy options, risk stratification, adjuvant treatment, and the defined role of clinical pharmacists. Access to innovative medicines is further constrained by high costs and insufficient reimbursement.
Conclusions. Significant differences in approaches to RCC pharmacotherapy between Ukraine and other countries were identified, indicating the need to harmonize national protocols with international guidelines, expand access to innovative therapies, and implement a multidisciplinary approach with pharmacist involvement to improve the effectiveness and accessibility of RCC treatment in Ukraine
References
- Siegel, R. L., Miller, K. D., Jemal, A. (2020). Cancer statistics, 2020. CA: A Cancer Journal for Clinicians, 70 (1), 7–30. https://doi.org/10.3322/caac.21590
- Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., Bray, F. (2021). Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: A Cancer Journal for Clinicians, 71 (3), 209–249. https://doi.org/10.3322/caac.21660
- Capitanio, U., Bensalah, K., Bex, A., Boorjian, S. A., Bray, F., Coleman, J. et al. (2019). Epidemiology of Renal Cell Carcinoma. European Urology, 75 (1), 74–84. https://doi.org/10.1016/j.eururo.2018.08.036
- Alivand, S., Fattahi, F., Zarei, Z., Hosseinifard, M., Nouralishahi, A., Karimi Aliabadi, H. et al. (2023). Assessing global kidney cancer incidence and mortality rates according to population category by income levels in 2020: An ecological study. Journal of Renal Injury Prevention, 12 (1). https://doi.org/10.34172/jrip.2023.32243
- Cai, Q., Chen, Y., Qi, X., Zhang, D., Pan, J., Xie, Z. et al. (2020). Temporal trends of kidney cancer incidence and mortality from 1990 to 2016 and projections to 2030. Translational Andrology and Urology, 9 (2), 166–181. https://doi.org/10.21037/tau.2020.02.23
- Goldstein, D. A., Clark, J., Tu, Y., Zhang, J., Fang, F., Goldstein, R., Stemmer, S. M., Rosenbaum, E. (2017). A global comparison of the cost of patented cancer drugs in relation to global differences in wealth. Oncotarget, 8 (42), 71548–71555. https://doi.org/10.18632/oncotarget.17742
- International Agency for Research on Cancer. GLOBOCAN. Available at: https://gco.iarc.fr/en
- Rak nyrky. Klinichna nastanova, zasnovana na dokazakh (2022). Derzhavnyi ekspertnyi tsentr ministerstva okhorony zdorovia Ukrainy. Available at: https://www.dec.gov.ua/wp-content/uploads/2022/06/2022_04_12-kn-rak-nyrky-2.pdf
- Powles, T., Albiges, L., Bex, A., Comperat, E., Grünwald, V., Kanesvaran, R. et al. (2024). Renal cell carcinoma: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Annals of Oncology, 35 (8), 692–706. https://doi.org/10.1016/j.annonc.2024.05.537
- Serzan, M. (2024). Management of Renal Cell Carcinoma. Journal of the National Comprehensive Cancer Network, 22. https://doi.org/10.6004/jnccn.2024.5011
- Larroquette, M., Peyraud, F., Domblides, C., Lefort, F., Bernhard, J.-C., Ravaud, A., Gross-Goupil, M. (2021). Adjuvant therapy in renal cell carcinoma: Current knowledges and future perspectives. Cancer Treatment Reviews, 97, 102207. https://doi.org/10.1016/j.ctrv.2021.102207
- Pontes, O., Oliveira-Pinto, S., Baltazar, F., Costa, M. (2022). Renal cell carcinoma therapy: Current and new drug candidates. Drug Discovery Today, 27 (1), 304–314. https://doi.org/10.1016/j.drudis.2021.07.009
- Mohammadi, M., Najafi, H., Mohammadi, P. (2025). CAR T-cell therapy in renal cell carcinoma: opportunities, challenges, and new strategies to overcome. Medical Oncology, 42 (6). https://doi.org/10.1007/s12032-025-02735-z
- El Zarif, T., Semaan, K., Xie, W., Eid, M., Zarba, M., Issa, W. et al. (2024). First-line Systemic Therapy Following Adjuvant Immunotherapy in Renal Cell Carcinoma: An International Multicenter Study. European Urology, 86 (6), 503–512. https://doi.org/10.1016/j.eururo.2024.07.016
- Wang, Y., Suarez, E. R., Kastrunes, G., de Campos, N. S. P., Abbas, R., Pivetta, R. S. et al. (2024). Evolution of cell therapy for renal cell carcinoma. Molecular Cancer, 23 (1). https://doi.org/10.1186/s12943-023-01911-x
- Gong, H., Ong, S. C., Li, F., Shen, Y., Weng, Z., Zhao, K. et al. (2024). Cost-effectiveness of immune checkpoint inhibitors as a first-line therapy for advanced hepatocellular carcinoma: a systematic review. Health Economics Review, 14 (1). https://doi.org/10.1186/s13561-024-00526-2
- Philip, E. J., Zhang, S., Tahir, P., Kim, D., Wright, F., Bell, A., Borno, H. T. (2021). Cost-Effectiveness of Immunotherapy Treatments for Renal Cell Carcinoma: A Systematic Review. Kidney Cancer, 5 (1), 47–62. https://doi.org/10.3233/kca-200107
- Wilson, B. E., Sullivan, R., Peto, R., Abubakar, B., Booth, C., Werutsky, G. et al. (2023). Global Cancer Drug Development – A Report From the 2022 Accelerating Anticancer Agent Development and Validation Meeting. JCO Global Oncology, 9. https://doi.org/10.1200/go.23.00294
- Pramesh, C. S., Badwe, R. A., Bhoo-Pathy, N., Booth, C. M., Chinnaswamy, G., Dare, A. J. et al. (2022). Priorities for cancer research in low- and middle-income countries: a global perspective. Nature Medicine, 28 (4), 649–657. https://doi.org/10.1038/s41591-022-01738-x
- Adunlin, G., Dong, J., Freeman, M. K. (2019). Immuno-Oncology Medicines: Policy Implications and Economic Considerations. Innovations in Pharmacy, 10 (3), 9. https://doi.org/10.24926/iip.v10i3.1799
- D’Angelo, S. P., Bhatia, S., Brohl, A. S., Hamid, O., Mehnert, J. M., Terheyden, P. et al. (2020). Avelumab in patients with previously treated metastatic Merkel cell carcinoma: long-term data and biomarker analyses from the single-arm phase 2 JAVELIN Merkel 200 trial. Journal for ImmunoTherapy of Cancer, 8 (1), e000674. https://doi.org/10.1136/jitc-2020-000674
- Zub, V. O., Kotuza, A. S. (2023). Analysis of providing care to patients with oncological diseases in the conditions of martial law. Bulletin of Social Hygiene and Health Protection Organization of Ukraine, 3, 35–40. https://doi.org/10.11603/1681-2786.2022.3.13434
- Svitovyi, O. (2023). Some aspects of functioning the pharmaceutical market of Ukraine. Economy and Society, 47. https://doi.org/10.32782/2524-0072/2023-47-80
- Fundamentals of the Legislation of Ukraine on Health Care (1992). Law of Ukraine No. 2801-XII. 19.11.1992. Available at: https://zakon.rada.gov.ua/laws/show/2801-12?lang=en#Text
- Pro systemu hromadskoho zdorovia (2022). Zakon Ukrainy No. 2573-IX. 06.09.2022. Available at: https://zakon.rada.gov.ua/laws/show/2573-20#Text
- On Public Procurement (2015). Law of Ukraine No. 922-VIII. 25.12.2015. Available at: https://zakon.rada.gov.ua/laws/show/922-19#Text
- On State Financial Guarantees of Medical Service to the Population (2017). Law of Ukraine No. 2168-VIII. 19.10.2017. Available at: https://zakon.rada.gov.ua/laws/show/2168–19#Text
- Pro skhvalennia Kontseptsii rozvytku elektronnoi okhorony zdorovia (2020). Rozporiadzhennia Kabinetu Ministriv Ukrainy No. 1671-r. 28.12.2020. Available at: https://zakon.rada.gov.ua/laws/show/1671-2020-%D1%80#Text
- Pro skhvalennia Natsionalnoi stratehii kontroliu zloiakisnykh novoutvoren na period do 2030 roku ta zatverdzhennia planu dii z yii realizatsii na period do 2025 roku (2024). Rozporiadzhennia Kabinetu Ministriv Ukrainy No. 730-r. 02.08.2024. Available at: https://zakon.rada.gov.ua/laws/show/730-2024-%D1%80#Text
- Pro zatverdzhennia Polozhennia pro multydystsyplinarnu komandu pry zakladakh okhorony zdorovia, shcho nadaiut spetsializovanu medychnu dopomohu patsiientam iz onkolohichnymy zakhvoriuvanniamy (2024). Nakaz Ministerstva okhorony zdorovia Ukrainy No. 1680. 02.10.2024. Available at: https://zakon.rada.gov.ua/laws/show/z1548-24#Text
- Pro zatverdzhennia form zvitnosti ta instruktsii shchodo yikh zapovnennia (2024). Nakaz Ministerstva okhorony zdorovia Ukrainy No. 2111. 18.12.2024. Available at: https://zakon.rada.gov.ua/laws/show/z0014-25#Text
- Pro systemu onkolohichnoi dopomohy naselenniu Ukrainy (2013). Nakaz Ministerstva okhorony zdorovia Ukrainy No. 845. 01.10.2013. Available at: https://zakon.rada.gov.ua/laws/show/z0077-14#Text
- Pro zatverdzhennia Unifikovanoho klinichnoho protokolu pervynnoi, vtorynnoi (spetsializovanoi) ta tretynnoi (vysokospetsializovanoi) medychnoi dopomohy «Rak nyrky» (2022). Nakaz Ministerstva okhorony zdorovia Ukrainy No. 1061. 20.06.2022. Available at: https://www.dec.gov.ua/mtd/rak-nyrky/
- Choueiri, T. K., Motzer, R. J. (2017). Systemic Therapy for Metastatic Renal-Cell Carcinoma. New England Journal of Medicine, 376 (4), 354–366. https://doi.org/10.1056/nejmra1601333
- Geynisman, D. M., Maranchie, J. K., Ball, M. W., Bratslavsky, G., Singer, E. A. (2021). A 25 year perspective on the evolution and advances in an understanding of the biology, evaluation and treatment of kidney cancer. Urologic Oncology: Seminars and Original Investigations, 39 (9), 548–560. https://doi.org/10.1016/j.urolonc.2021.04.038
- Rini, B. I., Campbell, S. C., Escudier, B. (2009). Renal cell carcinoma. The Lancet, 373 (9669), 1119–1132. https://doi.org/10.1016/s0140-6736(09)60229-4
- Lenvatinib in combination with Everolimus. U.S. Food and Drug Administration. Available at: https://www.fda.gov/drugs/resources-information-approved-drugs/lenvatinib-combination-everolimus
- Rebuzzi, S. E., Signori, A., Banna, G. L. L., Buti, S., Rescigno, P., Gemelli, M. et al. (2022). The prognostic role of nephrectomy in patients (pts) with metastatic renal cell carcinoma (mRCC) treated with immunotherapy according to the novel prognostic Meet-URO score: Subanalysis of the Meet-URO 15 study. Journal of Clinical Oncology, 40 (16), 4535–4535. https://doi.org/10.1200/jco.2022.40.16_suppl.4535
- Deiaki pytannia derzhavnoho rehuliuvannia tsin na likarski zasoby i vyroby medychnoho pryznachennia (2009). Postanova Kabinetu Ministriv Ukrainy No. 333. 25.03.2009. Available at: https://zakon.rada.gov.ua/laws/show/333-2009-%D0%BF?lang=en#Text
- Pro zatverdzhennia Pereliku likarskykh zasobiv, yaki pidliahaiut reimbursatsii za prohramoiu derzhavnykh harantii medychnoho obsluhovuvannia naselennia, stanom na 23 hrudnia 2024 roku (2024). Nakaz Ministerstva okhorony zdorovia Ukrainy No. 2148. 25.12.2024. Available at: https://zakon.rada.gov.ua/rada/show/v2148282-24#Text
- Powles, T., Tomczak, P., Park, S. H., Venugopal, B., Ferguson, T., Symeonides, S. N. et al. (2022). Pembrolizumab versus placebo as post-nephrectomy adjuvant therapy for clear cell renal cell carcinoma (KEYNOTE-564): 30-month follow-up analysis of a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. The Lancet Oncology, 23 (9), 1133–1144. https://doi.org/10.1016/s1470-2045(22)00487-9
- Mori, K., Yanagisawa, T., Fukuokaya, W., Iwatani, K., Matsukawa, A., Katayama, S. et al. (2023). Adjuvant immunotherapy in patients with renal cell carcinoma and urothelial carcinoma: A systematic review and network meta‐analysis. International Journal of Urology, 31 (1), 25–31. https://doi.org/10.1111/iju.15319
- Voylenko, O. A., Stakhovskyi, O. E., Vitruk, Y. V., Kononenko, O. A., Pikul, M. V., Grechko, B. O. et al. (2022). Neoadjuvant targeted therapy as a new approach to the treatment of patients with localized renal cell carcinoma. Clinical Oncology, 12 (3-4), 1–6. https://doi.org/10.32471/clinicaloncology.2663-466x.47-3.29236
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Oleksandr Kaiota, Alina Volkova, Natalia Khokhlenkova

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






