The frigoprotective effects of etoricoxib and diclofenac sodium in a model of acute general cooling in rats: the role of leukotrienes, interleukins, and nitric oxide synthase
DOI:
https://doi.org/10.15587/2519-4852.2026.359158Keywords:
frigoprotectors, acute general cooling, leukotrienes, interleukins, nitric oxide synthase, etoricoxib, diclofenac sodiumAbstract
The aim: The aim of the study was to evaluate the role of leukotrienes, interleukins, and nitric oxide synthase in frigoprotective effect of etoricoxib and diclofenac sodium in a model of acute general cooling in rats.
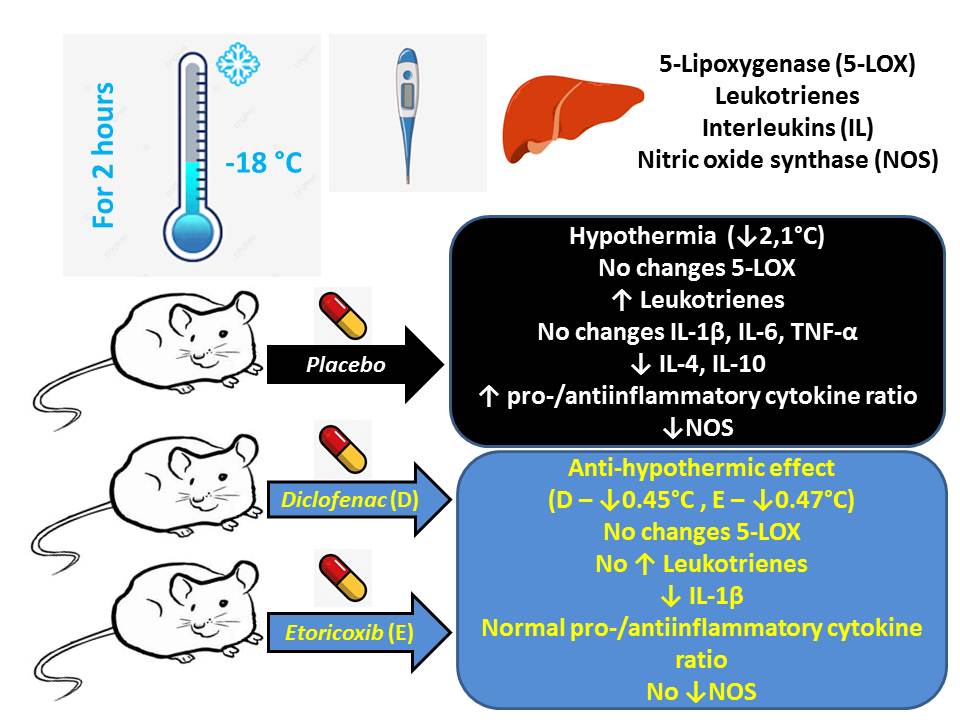
Material and methods: Acute general cooling was induced by exposing rats to −18 °C for 2 hours without animal mobility restriction. 30 min before cold exposure animals were treated with etoricoxib (5 mg/kg) or diclofenac sodium (7 mg/kg). Body temperature was measured before and after acute general cooling modeling. In rat liver the following parameters were determined: 5-lipoxygenase (5-LOX), leukotriene B4 (LTB4), total leukotrienes (LTs), interleukins (IL-1β, IL-4, IL-6, IL-10), tumor necrosis factor-α (TNF-α), and nitric oxide synthase (NOS).
Results: activation of the lipoxygenase pathway of arachidonic acid metabolism was characterized by a significant increase in leukotriene levels (total and leukotriene B4) without substantial changes in proinflammatory cytokines (IL-1β, IL-6, TNF-α) but with a significant decrease in anti-inflammatory cytokines (IL-4, IL-10) in the liver. Etoricoxib and diclofenac sodium similarly reduced the severity of hypothermia, prevented the increase in leukotriene levels without affecting 5-LOX content. Etoricoxib and particularly diclofenac sodium significantly reduced IL-1β levels without substantial changes in the other cytokines. Both studied medicines restored NOS levels to those observed in the intact control group.
Conclusions: obtained results experimentally substantiate the possibility of diclofenac sodium and etoricoxib to decrease the severity of acute general cooling, while also demonstrating certain differences in the mechanisms underlying their frigoprotective effects
Supporting Agency
- “Experimental substantiation for improving the effectiveness of prevention and treatment of cold injuries” of the list of scientific studies of the Ministry of Health of Ukraine, carried out at the expense of the state budget of Ukraine No. 0120u102460 (Order of the Ministry of Health of Ukraine No. 2651 of 17.11.2020).
References
- Oh, M., Song, G., An, D., Ahn, Y. (2025). Results of the 2024–2025 winter cold injury surveillance. Public Health Weekly Report 2025, 18 (43), 1671–1687. https://doi.org/10.56786/PHWR.2025.18.43.3
- Heil, K., Thomas, R., Robertson, G., Porter, A., Milner, R., Wood, A. (2016). Freezing and non-freezing cold weather injuries: a systematic review. British Medical Bulletin, 117 (1), 79–93. https://doi.org/10.1093/bmb/ldw001
- Tomassini, L., Lancia, M., Gambelunghe, C., Ricchezze, G., Fedeli, P., Cingolani, M. et al. (2025). Immunohistochemical insights into hypothermia-related deaths: a systematic review. Forensic Science, Medicine and Pathology, 21 (3), 1358–1369. https://doi.org/10.1007/s12024-024-00934-0
- Xie, Z., Huang, Z., Luo, W., Du, W. (2025). Burden of environmental heat and cold exposure and its attributable risk factors in 204 countries and territories (1990–2021). European Journal of Medical Research, 30 (1). https://doi.org/10.1186/s40001-025-03263-2
- QuickStats: Percentage Distribution of Deaths Attributed to Excessive Cold or Hypothermia,* by Month – United States, 2023 (2025). MMWR. Morbidity and Mortality Weekly Report, 74 (6), 107. https://doi.org/10.15585/mmwr.mm7406a6
- Duong, H., Patel, G., Holt, C. A. (2024). Hypothermia (Nursing). Treasure Island (FL): StatPearls Publishing.
- Liu, M., Patel, V. R., Wadhera, R. K. (2025). Cold-Related Deaths in the US. Jama, 333 (5), 427–429. https://doi.org/10.1001/jama.2024.25194
- Paal, P., Pasquier, M., Darocha, T., Lechner, R., Kosinski, S., Wallner, B. et al. (2022). Accidental Hypothermia: 2021 Update. International Journal of Environmental Research and Public Health, 19 (1), 501. https://doi.org/10.3390/ijerph19010501
- Frasch, J. J., König, H.-H., Konnopka, C. (2025). Effects of extreme temperature on morbidity, mortality, and case severity in German emergency care. Environmental Research, 270, 121021. https://doi.org/10.1016/j.envres.2025.121021
- Petersen, K. (2025). Accidental hypothermia in the elderly. Ugeskrift for Læger, 187 (24). https://doi.org/10.61409/v10240734
- Fourth winter of full-scale war leaves Ukrainians without doctors, medicines or warmth, new IRC data reveals. Kyiv: International Rescue Committee. Available at: https://www.rescue.org/press-release/fourth-winter-full-scale-war-leaves-ukrainians-without-doctors-medicines-or-warmth
- Jin, H.-X., Teng, Y., Dai, J., Zhao, X.-D. (2021). Expert consensus on the prevention, diagnosis and treatment of cold injury in China, 2020. Military Medical Research, 8 (1). https://doi.org/10.1186/s40779-020-00295-z
- Zhang, Y., Fu, M., Zhou, C., Wang, X., Jiang, Z., Jiang, C. et al. (2025). Cold exposure promotes the progression of osteoarthritis through downregulating APOE in cartilage. EMBO Molecular Medicine, 17 (8), 2137–2162. https://doi.org/10.1038/s44321-025-00268-6
- Sachs, C., Lehnhardt, M., Daigeler, A., Goertz, O. (2015). The Triaging and Treatment of Cold-Induced Injuries. Deutsches Ärzteblatt International, 112 (44), 741–747. https://doi.org/10.3238/arztebl.2015.0741
- Kapelka, I. G., Shtrygol, S. Y. (2019). The comparative research of frigoprotective properties of nonsteroidal anti-inflammatory drugs оn the model of acute general cooling. Pharmacology and Drug Toxicology, 13 (5), 338–343.
- Kapelka, I. G., Shtrygol, S. Y., Lesyk, R. B., Lozynskyi, A. V., Khomyak, S. V., Novikov, V. P. (2020). The comparative research of arachidonic acid cascade inhibitors for frigoprotective activity. Pharmacology and Drug Toxicology, 14 (2), 122–128. https://doi.org/10.33250/14.02.122
- Shtrygol’, S., Koiro, O., Kudina, O., Tovchiga, O., Yudkevych, T., Oklei, D. (2022). The influence of non-steroidal anti-inflammatory drugs with different mechanisms of action on the course of stress reaction, the functional state of kidneys, liver, and heart on the model of acute general cooling. ScienceRise: Pharmaceutical Science, 2 (36), 46–55. https://doi.org/10.15587/2519-4852.2022.255797
- Shtrygol’, S., Taran, A., Yudkevych, T., Lytkin, D., Lebedinets, I., Chuykova, P., Koiro, O. (2023). Effects of non-steroidal anti-inflammatory agents on systemic hemostasis during the most acute period of cold injury in rats. ScienceRise: Pharmaceutical Science, 6 (46), 25–30. https://doi.org/10.15587/2519-4852.2023.294311
- Shtrygol’, S., Tovchiga, O., Kudina, O., Koiro, O., Yudkevich, T., Gorbach, T. (2022). The effect of non-steroidal anti-inflammatory drugs with different mechanisms of action on the body temperature and cyclooxygenase pathway of the arachidonic acid cascade on the model of acute general cooling (air hypothermia) in rats. Česká a Slovenská Farmacie, 71 (5), 214–223. https://doi.org/10.5817/csf2022-5-214
- Kapelka, I. G., Shtrygol, S. Y., Koiro, O. O., Merzlikin, S. I., Kudina, O. V., Yudkevich, T. K. (2021). Effect of arachidonic acid cascade inhibitors on body temperature and cognitive functions in rats in the Morris water maze after acute cold injury. Die Pharmazie, 76 (7), 313–316. https://doi.org/10.1691/ph.2021.1571
- Voloshchuk, N. I., Yukhymchuk, A. V. (2023). Sex peculiarities of survival of animals with acute cold injury and correction with glucosamine hydrochloride. Pharmacology and Drug Toxicology, 17 (4), 248–254. https://doi.org/10.33250/17.04.248
- Bondariev, Y. V., Shtrygol’, S. Y., Drogovoz, S. M., Shchokina, K. G. (2018). Cold injury: preclinical study of drugs with frigoprotective properties. Kharkiv, 35.
- Yuhimchuk, A. V., Voloshchuk, N. I., Shtrygol, S. Yu., Nefodov, O. O., Piliponova, V. V., Oliinyk, Yu. M. et al. (2023). Vascular mechanisms in the formation of gender differences in the protective effect of glucosamine in experimental cold injury. World of Medicine and Biology, 19 (86), 243–247. https://doi.org/10.26724/2079-8334-2023-4-86-243-247
- Koval, A. Y., Strygol, S. Y., Lesyk, R. B., Lytkin, D. V., Lebedynets, I. O., Yudkevich, T. K. (2024). Frigoprotective properties of 5,7-diacyl-3-h(alkyl)-6-aryl-5h-[1,2,4]triazol[3,4-b][1,3,4]thiadiazine derivatives in the experiment. Medical and Clinical Chemistry, 3, 22–32. https://doi.org/10.11603/mcch.2410-681X.2024.i3.14912
- Eimonte, M., Eimantas, N., Daniuseviciute, L., Paulauskas, H., Vitkauskiene, A., Dauksaite, G. et al. (2021). Recovering body temperature from acute cold stress is associated with delayed proinflammatory cytokine production in vivo. Cytokine, 143,155510. https://doi.org/10.1016/j.cyto.2021.155510
- Vasek, D., Holicek, P., Galatik, F., Kratochvilova, A., Porubska, B., Somova, V. et al. (2024). Immune response to cold exposure: Role of γδ T cells and TLR2‐mediated inflammation. European Journal of Immunology. https://doi.org/10.1002/eji.202350897
- Russwurm, S., Stonāns, I., Schwerter, K., Stonāne, E., Meissner, W., Reinhart, K. (2002). Direct Influence of Mild Hypothermia on Cytokine Expression and Release in Cultures of Human Peripheral Blood Mononuclear Cells. Journal of Interferon & Cytokine Research, 22 (2), 215–221. https://doi.org/10.1089/107999002753536185
- Yuhimchuk, A. V., Voloshchuk, N. I. (2023). Influence of glucozamine on coagulation end aggregative hemostasis in male and female rats with acute cold injury. Prospects and innovations of science, 16 (34), 1024–1035. https://doi.org/10.52058/2786-4952-2023-16(34)-1024-1035
- Voloshchuk, N. I., Yuhimchuk, A. V. (2023). Gender dimorphism of changes in the skin microcirculation of rats on the background of acute cold trauma. Experimental and Clinical Physiology and Biochemistry, 68 (4), 21–27. https://doi.org/10.25040/ecpb2023.04.021
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Sergiy Shtrygol’, Olesia Kudina, Dmytro Lytkin, Andrii Taran, Tetiana Yudkevych

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






