Optimization of HPLC method for control of propranolol hydrochloride impurities in liquid medicine for oral use in pediatrics
DOI:
https://doi.org/10.15587/2519-4852.2026.350197Keywords:
verification, liquid chromatography, related substances, propranolol hydrochloride, small-scale manufacturing, infantile hemangiomaAbstract
Propranolol hydrochloride is approved in Ukraine as one of the drugs for the treatment of infantile hemangiomas in pediatrics, however, there are no industrially produced drugs, which necessitates the development and small-scale production. To ensure their proper quality, it is important to implement high-precision analytical control methods, to determine accompanying impurities that may affect the safety and effectiveness of therapy in children.
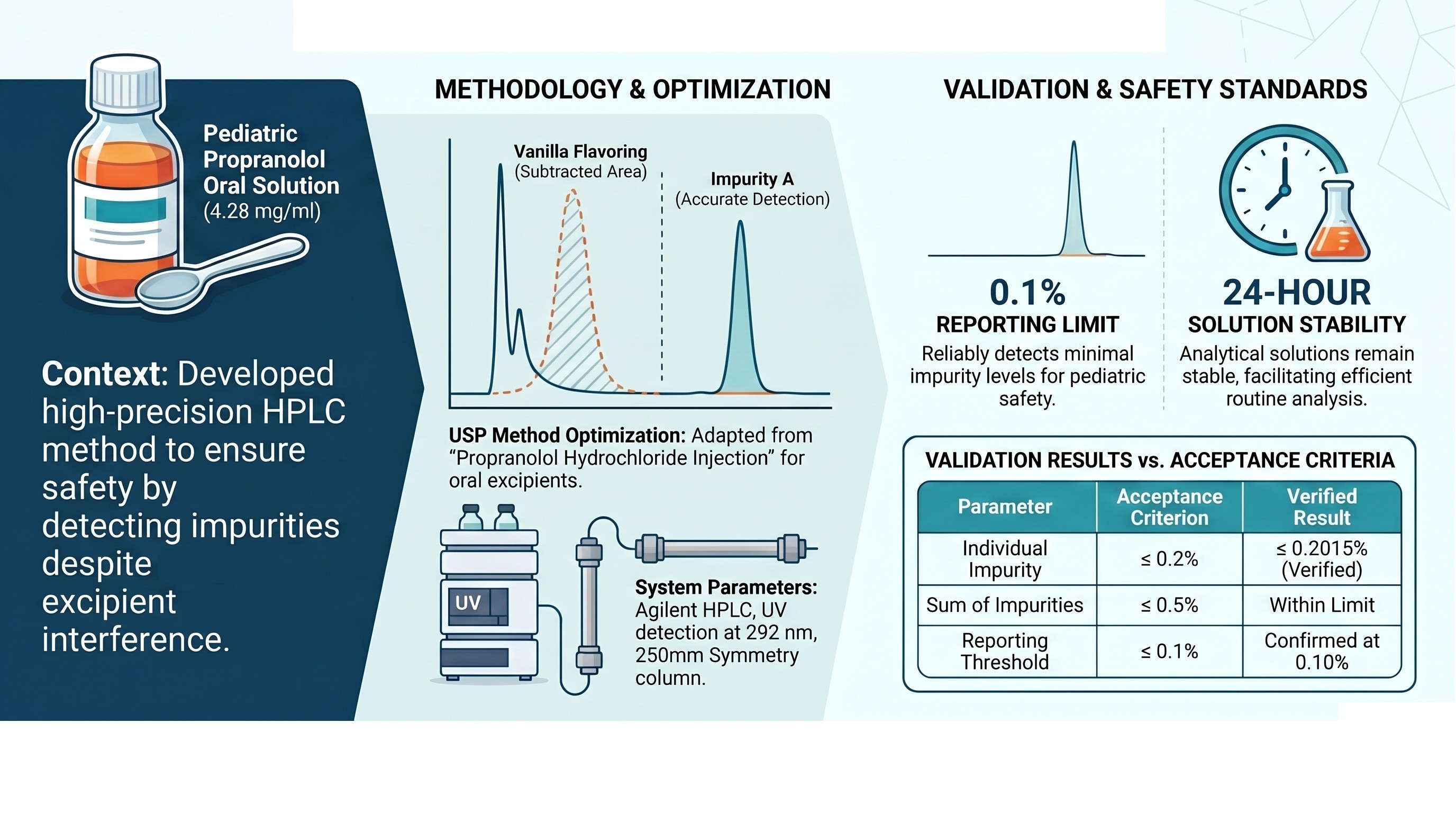
Aim. To optimize the liquid chromatography method for determining impurities of the active pharmaceutical ingredient in the drug "Propranolol hydrochloride, 4.28 mg/ml, oral solution", manufactured by pharmacy No. 2 CHEMOTEKA of the Pharmaceutical Department of PE "Infusia", intended for use in pediatrics.
Materials and methods. The study was carried out as part of the development of the medicinal product “Propranolol hydrochloride, 4.28 mg/ml, solution for oral administration” (pharmacy No. 2 CHEMOTEKA, PE "Infusia"). Impurities were controlled by liquid chromatography according to the US Pharmacopoeia method “Propranolol hydrochloride for injection”. Method verification was carried out considering the requirements of international standards and the SPhU.
Results. The proposed method for determining the content of related impurities is characterized by high specificity, linearity in the range of 0.5 - 1.6 mg/ml, accuracy, precision, confirmed reproducibility and reporting limit at the level of no more than 0.1%. Additionally, the stability of solutions for 24 hours has been established, which ensures the convenience of its use in routine pharmaceutical analysis.
Conclusions. The optimized liquid chromatography technique provides reliable determination of impurities in the medicinal product "Propranolol hydrochloride, 4.28 mg/ml, oral solution", meets international validation requirements and can be used in routine quality control. The results obtained confirm the feasibility of its use to improve the safety and effectiveness of therapy with the study drug
References
- Wu, Z., Zou, Y., Yuan, H., Fu, R., Jin, P. (2025). Efficacy and safety of propranolol tablets vs. oral solution in neonates with severe infantile hemangiomas: a retrospective study in China. Scientific Reports, 15 (1). https://doi.org/10.1038/s41598-024-84653-0
- Surzhykov, I. O., Bevz, O. V., Goncharuk, V. A., Kryvanych, O. V., Bevz, N. Y., Georgiyants, V. A. (2025). Prospects for off-label use of propranolol for the treatment of infantile hemangioma in children. Medychni Perspektyvy, 30 (3), 200–210. https://doi.org/10.26641/2307-0404.2025.3.340764
- Karunarathna, I., Lakmali, W., Hapuarachchi, T., Dissanayake, D. D., Gunasena, P., Aluthge, P. et al. (2024). The clinical use of propranolol: Mechanism, adverse effects, and interprofessional care. Uva Clinical Anaesthesia and Intensive Care, 7.
- CHMP assessment report: Hemangiol. International non-proprietary name: propranolol. Procedure No: EMEA/H/C/002621/0000 (2014). Committee for Medicinal Products for Human Use. London: European Medicines Agency. Available at: https://www.ema.europa.eu/en/documents/assessment-report/hemangiol-epar-public-assessment-report_en.pdf Last accessed: 09.09.2024
- Unifikovanyi klinichnyi protokol pervynnoi, vtorynnoi (spetsializovanoi) ta tretynnoi (vysokospetsializovanoi) medychnoi dopomohy. Sudynni anomalii u ditei (2016). Nakaz Ministerstva okhorony zdorovia Ukrainy No. 813. 08.08.2016. Available at: https://www.dec.gov.ua/wp-content/uploads/2019/11/2016_813_ykpmd_sa.pdf
- Serreau, R., Amirouche, A., Benyamina, A., Berteina-Raboin, S. (2024). Propranolol Hydrochloride Psychiatric Effectiveness and Oxidative Stress: An Update. Oxygen, 4 (2), 139–149. https://doi.org/10.3390/oxygen4020009
- Propranolol Hydrochloride (2020). The British Pharmacopoeia Vol. II, 735.
- Propranolol Hydrochloride. USP42-NF37. Available at: https://online.uspnf.com/uspnf/document/1_GUID-6E6003B7-D22E-4DA0-AE56-A389ACA7E54A_5_en-US
- Propranololu hidrokhloryd (2014). Derzhavna Farmakopeia Ukrainy. Vol. 2. Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 567–568.
- Propranolol Tablets (2020). The British Pharmacopoeia. Vol. III, 1182.
- Propranolol Hydrochloride Tablets. USP42-NF37. Available at: https://online.uspnf.com/uspnf/document/1_GUID-7ECACFAE-E9EC-47BF-A51C-35A7FD67EFC1_2_en-US
- Propranololu tabletky (2014). Derzhavna Farmakopeia Ukrainy. Vol. 3. Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 643–644.
- Propranolol Hydrochloride Injection. USP42-NF37. Available at: https://online.uspnf.com/uspnf/document/1_GUID-CB357EBF-84E9-44CF-9C07-281B09C2CF89_4_en-US
- USP42-NF37. Available at: https://online.uspnf.com/
- PA/PH/OMCL (13) 82 R5 – Validation/Verification of Analytical Procedures (2000). General European OMCL Network (GEON) Quality Management Document. Available at: https://www.edqm.eu/documents/52006/128968/omcl-validation-verification-of-analytical-procedures-paphomcl1382r5.pdf/5bd682ee-6c62-a352-c6ad-4cb2c31c749d?t=1628491790975
- Derzhavna Farmakopeia Ukrainy. Dopovnennia 7. Vol. 2 (2024). Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 424.
- 2.29. Ridynna khromatohrafiia (2025). Derzhavna Farmakopeia Ukrainy. Dopovnennia 5. Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 74–77.
- European Pharmacopoeia 12th ed. Available at: https://www.edqm.eu/en/european-pharmacopoeia-new-online-only-12th-edition
- Saito, J., Akabane, M., Ishikawa, Y., Iwahashi, K., Nakamura, H., Yamatani, A. (2020). Retrospective survey of compounded medications for children in Japan. European Journal of Pharmaceutics and Biopharmaceutics, 155, 122–127. https://doi.org/10.1016/j.ejpb.2020.08.016
- Saito, J., Yamatani, A., Kojima, Y., Nakakuni, M., Nakano, K., Hirose, K. et al. (2025). Stability and Dissolution Behavior Changes After Drug Compounding for Pediatric Cardiovascular Pharmacotherapy. Pharmaceutics, 17 (4), 535. https://doi.org/10.3390/pharmaceutics17040535
- Belayneh, A., Tessema, Z. (2021). A Systematic Review of the Stability of Extemporaneous Pediatric Oral Formulations. The Scientific World Journal, 2021, 1–9. https://doi.org/10.1155/2021/8523091
- Chatpalliwar, V., Bhavar, G. (2008). Quantitative analysis of propranolol hydrochloride by high performance thin layer chromatography. Indian Journal of Pharmaceutical Sciences, 70 (3), 395–398. https://doi.org/10.4103/0250-474x.43016
- Klovrzová, S., Zahálka, L., Matysová, L., Horák, P., Sklubalová, Z. (2013). Pediatric oral solutions with propranolol hydrochloride for extemporaneous compounding: the formulation and stability study. Česká a Slovenská Farmacie, 62 (1), 35–39. Available at: https://csfarmacie.cz/pdfs/csf/2013/01/07.pdf
- Pasham, M., Haridasyam, S. B., Vadagam, N., Boppy, N. V. V. D. P., Chinnakadoori, S. R., Lakka, N. S. (2023). Separation and quantification of organic‐related impurities of beta‐adrenergic receptor blocking agent propranolol in pharmaceutical solid dosage forms: Impurity profiling using stability‐indicating HPLC method. Separation Science Plus, 7 (1). https://doi.org/10.1002/sscp.202300159
- Saxena, S., Dubey, B. K., Basedia, D. K., Jain, P. K., Shah, S., Thakur, V. S. (2024). HPLC method development and validation for the estimation of propranolol and flunarizine in combined dosage form. Panacea Journal of Pharmacy and Pharmaceutical Sciences, 13 (1), 16–24. Available at: https://internationaljournal.org.in/journal/index.php/pjpps/article/view/991
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Olena Bevz, Anastasiia Slotina, Olga Kryvanych, Dmytro Soldatov, Nataliia Bevz, Oleksandr Shmalko, Victoriya Georgiyants

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






