Formulation and characterization of ezetimibe nanoparticles for hyperlipidemia treatment
DOI:
https://doi.org/10.15587/2519-4852.2026.359581Keywords:
Ezetimibe, HPLC, sodium alginate, nanoparticlesAbstract
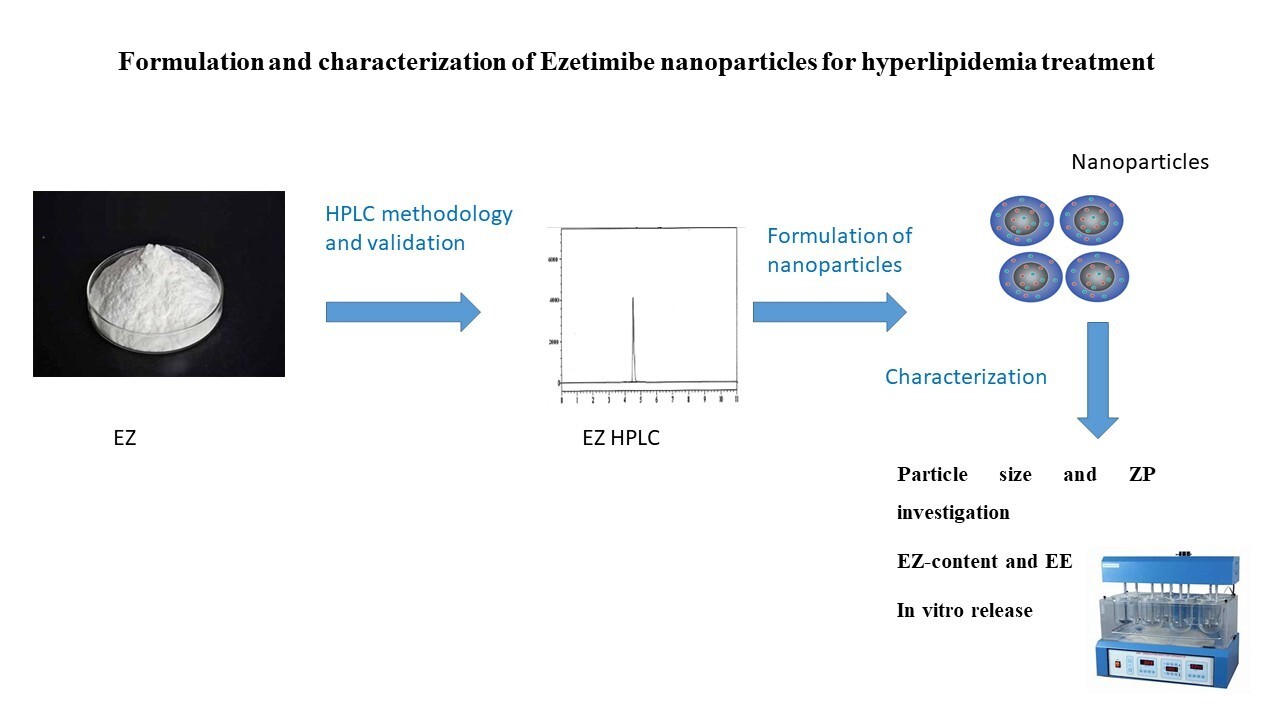
The aim of the work. Ezetimibe (EZ) is categorized as a Biopharmaceutics Classification System class II (BCS II) agent that possesses poor solubility and is highly permeable. This work aimed to incorporate EZ into nanoparticles (NPs) to accelerate release and to enhance its bioavailability.
Materials and Methods. A stability-indicating method for the computation of EZ was developed to compute EZ in NPs in fresh and stored samples. Nine formulations of EZ-NPs were developed by solvent evaporation using HPMC 6 cps, sodium alginate, and Tween 80 in various proportions. A HPLC method was designed to estimate EZ in NPs. EZ-NPs were tested chemically and characterized.
Results. The developed method was fully validated, and EZ NPs comprising HPMC and tween 80 had zeta potential (ZP) ranging from -21.6 mV to -30.1 mV, higher than other formulae, and their release was enhanced. The formulation (HP4) had an elevated ZP (-30.1 mV) and released about 91% of EZ within 20 min. HP4 was chosen for stability assessment and proved to be stable.
Conclusion. The formulation (HP4), including 0.1:0.5:0.4 ratios of the EZ: HPMC: sodium alginate, was the optimized EZ-NPs formulation. Microencapsulation of EZ with HPMC: sodium alginate in a 0.1:0.5:0.4 ratio could enhance the release and achieve stability
References
- Nițu, E.-T., Jianu, N., Merlan, C., Foica, D., Sbârcea, L., Buda, V. et al. (2025). A Comprehensive Review of the Latest Approaches to Managing Hypercholesterolemia: A Comparative Analysis of Conventional and Novel Treatments: Part I. Life, 15 (8), 1185. https://doi.org/10.3390/life15081185
- Kumari, L., Rani, D. (2025). A Concise overview of diverse analytical techniques for ezetimibe estimation. Microchemical Journal, 209, 112561. https://doi.org/10.1016/j.microc.2024.112561
- Torrado-Salmerón, C., Guarnizo-Herrero, V., Gallego-Arranz, T., del Val-Sabugo, Y., Torrado, G., Morales, J., Torrado-Santiago, S. (2020). Improvement in the Oral Bioavailability and Efficacy of New Ezetimibe Formulations – Comparative Study of a Solid Dispersion and Different Micellar Systems. Pharmaceutics, 12 (7), 617. https://doi.org/10.3390/pharmaceutics12070617
- Lipinski, C. A. (2000). Drug-like properties and the causes of poor solubility and poor permeability. Journal of Pharmacological and Toxicological Methods, 44 (1), 235–249. https://doi.org/10.1016/s1056-8719(00)00107-6
- Zhuo, Y., Zhao, Y.-G., Zhang, Y. (2024). Enhancing Drug Solubility, Bioavailability, and Targeted Therapeutic Applications through Magnetic Nanoparticles. Molecules, 29 (20), 4854. https://doi.org/10.3390/molecules29204854
- A. Ali, Y., N. Abd-Alhammid, S. (2017). Formulation and Evaluation of Ezetimibe Nanoparticles. Iraqi Journal of Pharmaceutical Sciences, 24 (2), 11–21. https://doi.org/10.31351/vol24iss2pp11-21
- Q 2 (R1) Validation of Analytical Procedures: Text and Methodology (1995). European Medicines Agency. Available at: https://www.ema.europa.eu/en/documents/scientific-guideline/ich-guideline-q2r1-validation-analytical-procedures-text-methodology-step-5-first-version_en.pdf
- The Pharmacopoeia of United States of America (2020). National Formulary 38, Mack publishing Co. Easton, vol. 2. Available at: http://182.160.97.198:8080/xmlui/handle/123456789/1493
- Singh, R., Popli, H., Suryawanshi, M. (2025). Development and validation of an HPLC analytical method for the simultaneous estimation of vitamin B12 and folic acid. Essential Chem, 2 (1), 1–9. https://doi.org/10.1080/28378083.2025.2462931
- Rostami, E. (2021). Recent achievements in sodium alginate-based nanoparticles for targeted drug delivery. Polymer Bulletin, 79 (9), 6885–6904. https://doi.org/10.1007/s00289-021-03781-z
- Filimon, A., Onofrei, M. D., Bargan, A., Stoica, I., Dunca, S. (2023). Bioactive Materials Based on Hydroxypropyl Methylcellulose and Silver Nanoparticles: Structural-Morphological Characterization and Antimicrobial Testing. Polymers, 15 (7), 1625. https://doi.org/10.3390/polym15071625
- Li, H.-J., Zhang, A.-Q., Hu, Y., Sui, L., Qian, D.-J., Chen, M. (2012). Large-scale synthesis and self-organization of silver nanoparticles with Tween 80 as a reductant and stabilizer. Nanoscale Research Letters, 7 (1). https://doi.org/10.1186/1556-276x-7-612
- Elhassan, E., Omolo, C. A., Gafar, M. A., Ismail, E. A., Ibrahim, U. H., Khan, R. et al. (2025). Multifunctional hyaluronic acid-based biomimetic/pH-responsive hybrid nanostructured lipid carriers for treating bacterial sepsis. Journal of Biomedical Science, 32 (1). https://doi.org/10.1186/s12929-024-01114-6
- Jafar, M., Sajjad Ahmad Khan, M., Salahuddin, M., Zahoor, S., MohammedHesham Slais, H., Ibrahim Alalwan, L., Radhi Alshaban, H. (2023). Development of apigenin loaded gastroretentive microsponge for the targeting of Helicobacter pylori. Saudi Pharmaceutical Journal, 31 (5), 659–668. https://doi.org/10.1016/j.jsps.2023.03.006
- Castro, S. R., Ribeiro, L. N. M., Breitkreitz, M. C., Guilherme, V. A., Rodrigues da Silva, G. H., Mitsutake, H. et al. (2021). A pre-formulation study of tetracaine loaded in optimized nanostructured lipid carriers. Scientific Reports, 11 (1). https://doi.org/10.1038/s41598-021-99743-6
- Dissolution Methods. FDA. Available at: https://www.accessdata.fda.gov/scripts/cder/dissolution/dsp_SearchResults.cfm
- Rahi, F. A., Mohammed Ameen, M. S., Fayyadh, M. S. (2021). Linagliptin and gliclazide di-loaded extended-release nanoparticles: formulation and evaluation. Wiadomości Lekarskie, 74 (9), 2315–2322. https://doi.org/10.36740/wlek202109212
- Rahi, F. A., Ameen, M. S. M., Jawad, K. K. M. (2022). Preparation and evaluation of lipid matrix microencapsulation for drug delivery of azilsartan kamedoxomil. ScienceRise: Pharmaceutical Science, 6 (40), 21–28. https://doi.org/10.15587/2519-4852.2022.270306
- Singh, N., Akhtar, M. J., Anchliya, A. (2021). Development and Validation of HPLC Method for Simultaneous Estimation of Reduced and Oxidized Glutathione in Bulk Pharmaceutical Formulation. Austin Journal of Analytical and Pharmaceutical Chemistry, 8 (1). https://doi.org/10.26420/austininternmed.2021.1129
- Sudha, T., Sumithra, M., Ajith, U. (2024). A review of analytical method development and validation of labetalol hydrochloride. Annals of Phytomedicine an International Journal, 13 (1). https://doi.org/10.54085/ap.2024.13.1.38
- Mahapatra, A., Sreedhar, C. (2018). HPLC and analytical method validation: a review. Indian Research Journal of Pharmacy and Science, 5 (1), 1399–1414. https://doi.org/10.21276/irjps.2018.5.1.18
- Nakatuka, Y., Yoshida, H., Fukui, K., Matuzawa, M. (2015). The effect of particle size distribution on effective zeta-potential by use of the sedimentation method. Advanced Powder Technology, 26 (2), 650–656. https://doi.org/10.1016/j.apt.2015.01.017
- Elkhayat, D., Abdelmalak, N. S., Amer, R., Awad, H. H. (2025). Ezetimibe Loaded Nanostructured Lipid Carriers Tablets: Response Surface Methodology, In-vitro Characterization, and Pharmacokinetics Study in Rats. Journal of Pharmaceutical Innovation, 20 (1). https://doi.org/10.1007/s12247-024-09911-0
- Tulain, U. R., Mahmood, A., Aslam, S., Erum, A., Shamshad Malik, N., Rashid, A. et al. (2021). Formulation and Evaluation of Linum usitatissimum Mucilage-Based Nanoparticles for Effective Delivery of Ezetimibe. International Journal of Nanomedicine, 16, 4579–4596. https://doi.org/10.2147/ijn.s308790
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Asmaa Abdelaziz Mohamed, Olla Maan, Firas Aziz Rahi, Doaa Zakaria Elkashif

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






