Research in component composition of essential oils from various organs of Silphium perfoliatum L.
DOI:
https://doi.org/10.15587/2519-4852.2026.352599Keywords:
Silphium perfoliatum L., Asteraceae, essential oil, component composition, GC/MSAbstract
Silphium perfoliatum L. (cup plant) is a perennial member of the Asteraceae family indigenous to eastern North America. Its phytochemical profile and biological properties have not yet been comprehensively characterised, and the species is not included in official medical practice. Nevertheless, ethnobotanical records indicate that Indigenous communities of North America traditionally used this plant to alleviate conditions such as neuralgia, respiratory ailments, and rheumatic disorders.
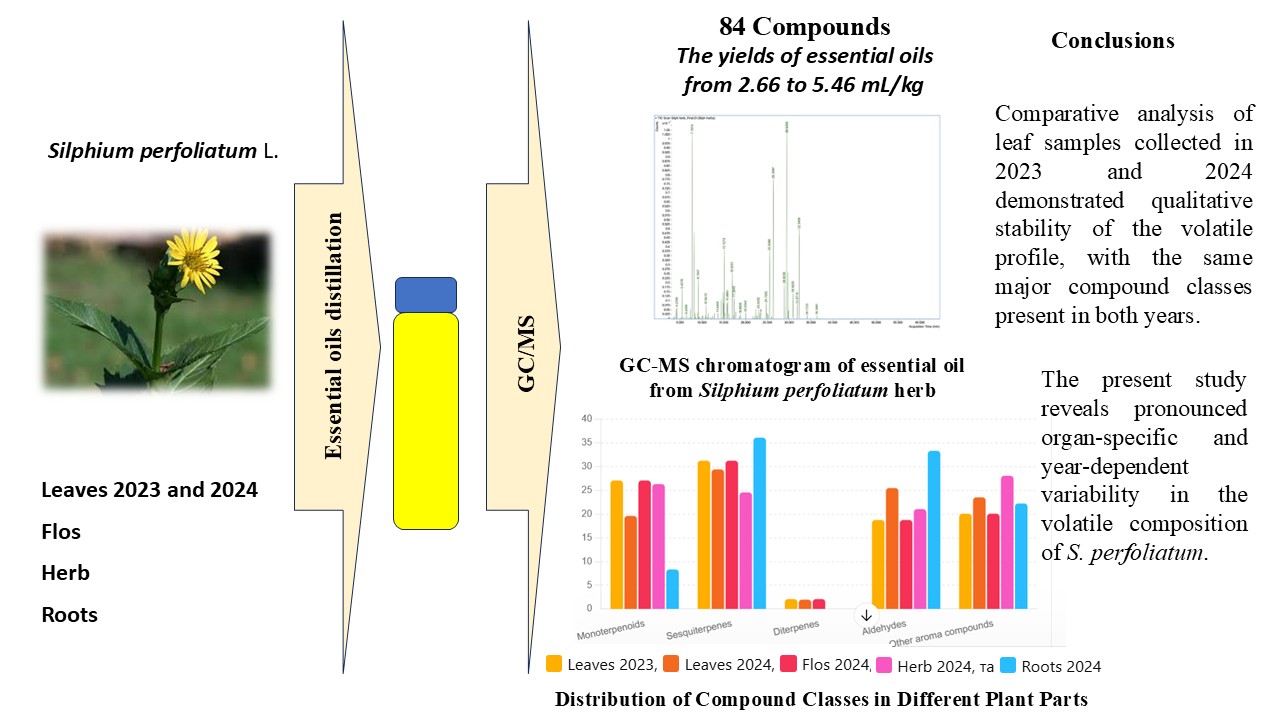
The aim. The aim of this study was to conduct a comprehensive analysis of the qualitative and quantitative composition of essential oils isolated from different organs of S. perfoliatum L. to identify organ-dependent variations and expand current knowledge on the phytochemical profile of this species.
Materials and methods. The plant materials of S. perfoliatum used in this study were collected in Ivano-Frankivsk, Ukraine. Hydrodistillates obtained from dried various organs of S. perfoliatum L. according to the methods of the European Pharmacopoeia were analyzed using gas chromatography coupled with mass spectrometric detection (GC–MS) using an Agilent 6890/5973 GC–MS system operated with ChemStation software for mass- selective detectors (MSD). Agilent HP-5MSI capillary column (30 m × 0.25 mm i.d., film thickness 0.25 µm) were used.
Research results. The yields of essential oils (EOs) in S. perfoliatum L. organs ranged from 2.66 to 5.46 mL/kg. In total, 84 volatile compounds were identified in the raw materials, including monoterpenoids, sesquiterpenes, diterpenes, aldehydes, and other aroma compounds. Sesquiterpenes were the dominant class in all samples (60.44–77.53%). Caryophyllene oxide, germacrene-type alcohols, and caryophyllene prevailed in the aerial parts, whereas root distillates were characterised by silphiperfol derivatives such as silphiperfol-5-ene and presilphiperfol-7-ene. Monoterpenes were most abundant in flowers (23.60%), with α-pinene and camphene as key constituents, while roots contained negligible amounts. Diterpenes, mainly phytol, were detected predominantly in leaves, highlighting organ-specific differences in volatile biosynthesis. A comparative analysis of leaf essential oils collected in 2023 and 2024 demonstrated qualitative stability with quantitative variation. Sesquiterpenes remained dominant in both years (67.19% in 2023; 60.51% in 2024). Caryophyllene oxide and germacrene-type alcohols were major constituents in both samples, though phytol content increased markedly in 2024 (16.54% vs 5.46%). In contrast, 2023 samples showed higher levels of monoterpenes (7.97% vs 5.88%) and aldehydes (7.72% vs 4.36%), indicating seasonal shifts in volatile composition.
Conclusions. This research elucidates the volatile profiles of S. perfoliatum from different organs and harvest years, demonstrating clear organ-related and interannual variability. The content of volatile fractions varied from 2.66 to 5.46 mL/kg, depending on the plant organs. A total of 84 volatile compounds were identified, with sesquiterpenes representing the dominant class in all samples (60.44–77.53%). Aerial parts were enriched in oxygenated sesquiterpenes such as caryophyllene oxide and germacrene-derived alcohols, whereas roots showed a distinct chemotype dominated by silphiperfol-type hydrocarbons. Monoterpenes were most abundant in the flowers (23.60%), with α-pinene, camphene, and oxygenated monoterpenes contributing substantially to the volatile profile, while their content in roots was negligible. Overall, these results broaden current understanding of the phytochemical diversity of S. perfoliatum and support further investigation of its essential oils for potential biological and applied uses
Supporting Agency
- European Union in the MSCA4Ukraine project “Design and development of 3D-printed medicines for bioactive materials of Ukrainian and Estonian medicinal plants origin” [ID number 1232466]
References
- Britton, N. L., Brown, A. (1913) An illustrated flora of the northern United States, Canada and the British possessions : from Newfoundland to the parallel of the southern boundary of Virginia, and from the Atlantic Ocean westward to the 102d meridian. New York . https://doi.org/10.5962/bhl.title.940
- Silphium perfoliatum. Flora of North America. Available at: http://www.efloras.org/florataxon.aspx?flora_id=1&taxon_id=242417268
- Albrecht, K. A., Goldstein, W. (1997). Silphium perfoliatum: A North American Prairie Plant with Potential as a Forage Crop. The XVIII International Grassland Congress. Winnipeg.
- Silphium perfoliatum. Royal Botanic Gardens. Available at: https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:236461-2
- Ende, L. M., Knöllinger, K., Keil, M., Fiedler, A. J., Lauerer, M. (2021). Possibly Invasive New Bioenergy Crop Silphium perfoliatum: Growth and Reproduction Are Promoted in Moist Soil. Agriculture, 11 (1), 24. https://doi.org/10.3390/agriculture11010024
- Kowalski, R., Wolski, T. (2003). Evaluation of phenolic acid content in Silphium perfoliatum L. leaves, inflorescences and rhizomes. Electronic Journal of Polish Agricultural Universities, 6 (1).
- Kowalski, R., Wolski, T. (2005). The chemical composition of essential oils of Silphium perfoliatum L. Flavour and Fragrance Journal, 20 (3), 306–310. https://doi.org/10.1002/ffj.1418
- Kowalski, R., Wierciński, J., Mardarowicz, M. (2005). Essential Oil in Leaves and Inflorescences ofSilphium integrifoliumMichx. Journal of Essential Oil Research, 17 (2), 220–222. https://doi.org/10.1080/10412905.2005.9698881
- Williams, J. D., Wojcińska, M., Calabria, L. M., Linse, K., Clevinger, J. A., Mabry, T. J. (2009). The Flavonoids and Phenolic Acids of the Genus Silphium and Their Chemosystematic Value. Natural Product Communications, 4 (3). https://doi.org/10.1177/1934578x0900400325
- Gilmore, M. R. (1919). Uses of plants by the Indians of the Missouri River region. Lincoln: University of Nebraska Press. https://doi.org/10.5962/bhl.title.32507
- Kowalski, R., Kędzia, B. (2007). Antibacterial Activity ofSilphium perfoliatum. Extracts. Pharmaceutical Biology, 45 (6), 494–500. https://doi.org/10.1080/13880200701389409
- Zhang, G., Jia, W., Liu, L., Wang, L., Xu, J., Tao, J. et al. (2025). Caffeoylquinic acids from Silphium perfoliatum L. show hepatoprotective effects on cholestatic mice by regulating enterohepatic circulation of bile acids. Journal of Ethnopharmacology, 337, 118870. https://doi.org/10.1016/j.jep.2024.118870
- Xu, J., Jia, W., Zhang, G., Liu, L., Wang, L., Wu, D. et al. (2024). Extract of Silphium perfoliatum L. improve lipid accumulation in NAFLD mice by regulating AMPK/FXR signaling pathway. Journal of Ethnopharmacology, 327, 118054. https://doi.org/10.1016/j.jep.2024.118054
- Dobrochaeva, D. N., Kotov, M. Y., Prokudyn, Yu. N., Barbarych, A. Y. (1999). Opredelytel vysshykh rastenyi Ukrayny. Kyiv: Naukova Dumka.
- EMEA/HMPC/246816/2005. Guideline on Good Agricultural and Collection Practice (GACP) for Starting Materials of Herbal Origin (2006). European Medicines Agency.
- European Pharmacopoeia (2022). Strasbourg: Council of Europe.
- Raal, A., Ilina, T., Kovalyova, A., Koshovyi, O. (2024). Volatile compounds in distillates and hexane extracts from the flowers of Philadelphus coronarius and Jasminum officinale. ScienceRise: Pharmaceutical Science, 6 (52), 37–46. https://doi.org/10.15587/2519-4852.2024.318497
- Raal, A., Dolgošev, G., Ilina, T., Kovalyova, A., Lepiku, M., Grytsyk, A., Koshovyi, O. (2025). The Essential Oil Composition in Commercial Samples of Verbena officinalis L. Herb from Different Origins. Crops, 5 (2), 16. https://doi.org/10.3390/crops5020016
- Raal, A., Liira, J., Lepiku, M., Ilina, T., Kovalyova, A., Strukov, P. et al. (2025). The Composition of Essential Oils and the Content of Saponins in Different Parts of Gilia capitata Sims. Crops, 5 (3), 33. https://doi.org/10.3390/crops5030033
- Hrytsyk, Y., Koshovyi, O., Lepiku, M., Jakštas, V., Žvikas, V., Matus, T. et al. (2024). Phytochemical and Pharmacological Research in Galenic Remedies of Solidago canadensis L. Herb. Phyton, 93 (9), 2303–2315. https://doi.org/10.32604/phyton.2024.055117
- Raal, A., Komarov, R., Orav, A., Kapp, K., Grytsyk, A., Koshovyi, O. (2022). Chemical composition of essential oil of common juniper (Juniperus communis L.) branches from Estonia. ScienceRise: Pharmaceutical Science, 6 (40), 66–73. https://doi.org/10.15587/2519-4852.2022.271048
- Sepp, J., Koshovyi, O., Jakštas, V., Žvikas, V., Botsula, I., Kireyev, I. et al. (2024). Phytochemical, Pharmacological, and Molecular Docking Study of Dry Extracts of Matricaria discoidea DC. with Analgesic and Soporific Activities. Biomolecules, 14 (3), 361. https://doi.org/10.3390/biom14030361
- Francomano, F., Caruso, A., Barbarossa, A., Fazio, A., La Torre, C., Ceramella, J. et al. (2019). β-Caryophyllene: A Sesquiterpene with Countless Biological Properties. Applied Sciences, 9 (24), 5420. https://doi.org/10.3390/app9245420
- Baradaran Rahimi, V., Askari, V. R. (2022). A mechanistic review on immunomodulatory effects of selective type two cannabinoid receptor β‐caryophyllene. BioFactors, 48 (4), 857–882. https://doi.org/10.1002/biof.1869
- Baradaran Rahimi, V., Memarzia, A., Askari, V. R. (2023). Antiinflammatory and antioxidant and immunomodulatory effects of phytocannabinoid β-caryophyllene: A mechanistic review. Medicinal Usage of Cannabis and Cannabinoids. Elsevier, 349–359. https://doi.org/10.1016/b978-0-323-90036-2.00010-7
- Salehi, B., Upadhyay, S., Erdogan Orhan, I., Kumar Jugran, A., L. D. Jayaweera, S., A. Dias, D. et al. (2019). Therapeutic Potential of α- and β-Pinene: A Miracle Gift of Nature. Biomolecules, 9 (11), 738. https://doi.org/10.3390/biom9110738
- Zielińska-Błajet, M., Feder-Kubis, J. (2020). Monoterpenes and Their Derivatives – Recent Development in Biological and Medical Applications. International Journal of Molecular Sciences, 21 (19), 7078. https://doi.org/10.3390/ijms21197078
- Cheekatla, S. R. (2024). Triquinane based natural products via cycloadditions and metathesis. Tetrahedron Chem, 12, 100103. https://doi.org/10.1016/j.tchem.2024.100103
- Qiu, Y., Lan, W.-J., Li, H.-J., Chen, L.-P. (2018). Linear Triquinane Sesquiterpenoids: Their Isolation, Structures, Biological Activities, and Chemical Synthesis. Molecules, 23 (9), 2095. https://doi.org/10.3390/molecules23092095
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Raal Ain, Liubomyr Lytvynets, Andriy Grytsyk, Ivan Bilai, Oleh Koshovyi

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






