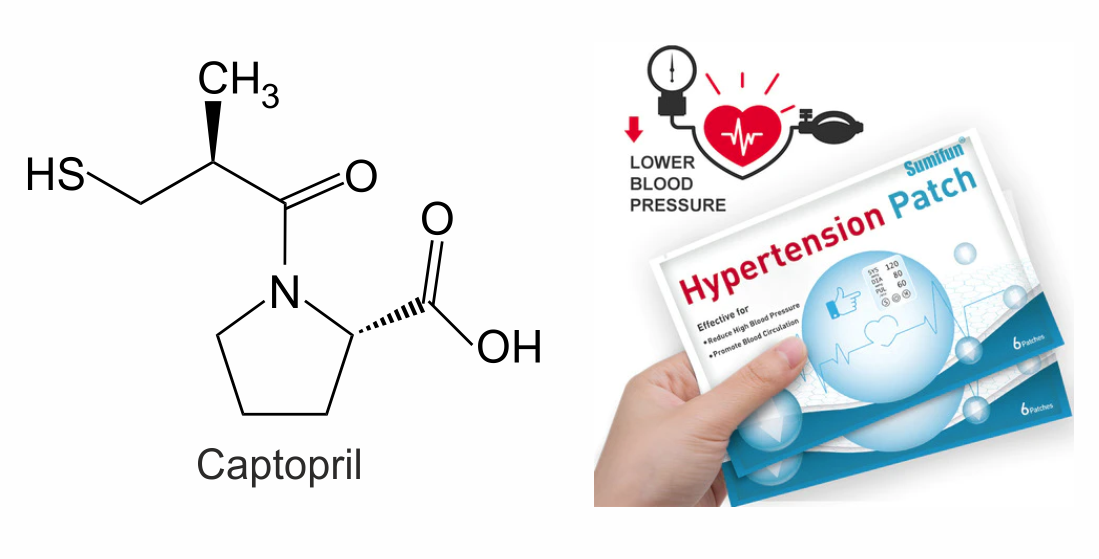
Преформуляційні дослідження з розробки транcдермальної терапевтичної системи з каптоприлом
DOI:
https://doi.org/10.15587/2519-4852.2026.353006Ключові слова:
гіпертензія, каптоприл, проникність in vitro, трансдермальна терапевтична система (ТТС)Анотація
У статті викладено результати дослідження процесу проникнення каптоприлу через напівпроникну мембрану шляхом діалізу in vitro для практичного підтвердження можливості його використання для створення нових форм доставки – трансдермальних терапевтичних систем (ТТС).
Мета: Преформуляційні дослідження фармацевтичної розробки трансдермальної лікарської форми ТТС антигіпертензивної дії з каптоприлом, визначення природи та кінетичних параметрів процесу проникності каптоприлу через напівпроникну мембрану in vitro, а також впливу початкової концентрації обраного активного фармацевтичного інгредієнта (АФІ) на цей процес.
Матеріали та методи: На початковому етапі розробки антигіпертензивної ТТС досліджували процес проникнення каптоприлу через напівпроникну мембрану шляхом діалізу in vitro при (37±0,5) °C. Як дифузійне середовище використовували фосфатний буферний розчин (pH 7,4). Початкова концентрація каптоприлу в донорному розчині становила 30 мг/мл.
Результати: На основі аналізу отриманих експериментальних значень кількості досліджуваної речовини у зразку діалізату Хi та питомого градієнта потоку за одиницю часу ΔQt було визначено, що процес проникнення каптоприлу в модельних умовах характеризується рівномірною швидкістю та відповідає кінетиці нульового порядку. Високе значення коефіцієнта кореляції R = 0,9996 для отриманого кінетичного рівняння підтверджує лінійну залежність проходження досліджуваної речовини через мембрану від часу.
Висновок: Проведені дослідження з визначення кількісних характеристик проникності показали, перш за все, здатність молекул обраної речовин долати мембранні бар’єри і дозволяють дати позитивну оцінку прийнятності цього активного фармацевтичного інгредієнта як привабливого для створення ТТС
Спонсор дослідження
- Ministry of Health of Ukraine from the state budget according to the topic «Preformulation studies of biopharmaceutical properties of active pharmaceutical ingredients with antihypertensive action for the creation of innovative transdermal drug delivery systems» (state registration number: 0122U001605).
Посилання
- Mills, K. T., Stefanescu, A., He, J. (2020). The global epidemiology of hypertension. Nature Reviews Nephrology, 16 (4), 223–237. https://doi.org/10.1038/s41581-019-0244-2
- Whelton, P. K., Carey, R. M., Aronow, W. S., Casey, D. E., Collins, K. J., Dennison Himmelfarb, C. et al. (2018). 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension, 71 (6), 1269–1324. https://doi.org/10.1161/hyp.0000000000000066
- Hypertension. World Health Organization. Available at: https://www.who.int/health-topics/hypertension/#tab=tab_1
- World Hypertension Day 2023. International Society of Hypertension. Available at: https://ish-world.com/world-hypertension-day-2023/
- Unger, T., Borghi, C., Charchar, F., Khan, N. A., Poulter, N. R., Prabhakaran, D. et al. (2020). 2020 International Society of Hypertension Global Hypertension Practice Guidelines. Hypertension, 75 (6), 1334–1357. https://doi.org/10.1161/hypertensionaha.120.15026
- Herman, L. L., Padala, S. A., Ahmed, I., Bashir, K. (2023). Angiotensin-Converting Enzyme Inhibitors (ACEI). StatPearls. StatPearls Publishing. Available at: https://pubmed.ncbi.nlm.nih.gov/28613705/
- Chen, Y. J., Li, L. J., Tang, W. L., Song, J. Y., Qiu, R., Li, Q. et al. (2018). First-line drugs inhibiting the renin angiotensin system versus other first-line antihypertensive drug classes for hypertension. Cochrane Database of Systematic Reviews, 2018 (11). https://doi.org/10.1002/14651858.cd008170.pub3
- Marte, F., Sankar, P., Patel, P., Cassagnol, M. (2024). Captopril. StatPearls. StatPearls Publishing. Available at: https://www.ncbi.nlm.nih.gov/books/NBK535386/
- Obied, A. H. H., Ahmed, A. A. E. (2021). Evaluation of the clinical outcome of captopril use for hypertensive urgency in Khartoum State’s emergency centres. African Journal of Emergency Medicine, 11 (1), 202–206. https://doi.org/10.1016/j.afjem.2020.10.003
- Captopril (Captoprilum). Compendium. Available at: https://compendium.com.ua/akt/67/3102/captoprilum/
- Shyteyeva, T., Bezchasnyuk, E., Kryskiv, O., Grynenko, V. (2023). Substantiation of creation of transdermal forms of drug delivery with antihypertensive action. ScienceRise: Pharmaceutical Science, 4 (44), 104–113. https://doi.org/10.15587/2519-4852.2023.286303
- Pastore, M. N., Kalia, Y. N., Horstmann, M., Roberts, M. S. (2015). Transdermal patches: history, development and pharmacology. British Journal of Pharmacology, 172 (9), 2179–2209. https://doi.org/10.1111/bph.13059
- Patches, transdermal. (2016) Ph. Eur. 10.0. Strasbourg: Council of Europe, 925. Available at: https://www.scribd.com/document/508063535/European-Pharmacopoeia-10-0#page=957
- Vachhal, I. K., Kumar, K., Joshi, A., Rajput, V. (2023). Transdermal patches: updated review as a novel drug delivery system. UPI Journal of Pharmaceutical, Medical and Health Sciences, 6 (4), 7–11. https://doi.org/10.37022/jpmhs.v6i4.95
- Xu, F., Qiu, Z., Zhang, M., Ren, Y., Kong, L., Liu, Y. et al. (2025). Transdermal Drug Delivery Systems: A Comprehensive Review of Mechanisms, Technologies, and Clinical Applications. Pharmaceutical Research, 42 (12), 2429–2442. https://doi.org/10.1007/s11095-025-03962-9
- Crasta, A., Painginkar, T., Sreedevi, A., Pawar, S. D., Badamane Sathyanarayana, M. et al. (2025). Transdermal drug delivery system: A comprehensive review of innovative strategies, applications, and regulatory perspectives. OpenNano, 24, 100245. https://doi.org/10.1016/j.onano.2025.100245
- Lee, H., Song, C., Baik, S., Kim, D., Hyeon, T., Kim, D.-H. (2018). Device-assisted transdermal drug delivery. Advanced Drug Delivery Reviews, 127, 35–45. https://doi.org/10.1016/j.addr.2017.08.009
- Global Transdermal Drug Delivery Market Trends and Drivers, Restraints, and Opportunities 2017–2023 (2018). Dublin. PRNewswire. Available at: https://www.prnewswire.com/news-releases/global-transdermal-drug-delivery-market-2017-2023-key-players-are-3m-boehringer-ingelheim-johnson-and-johnson-mylan-nv-novartis-and-glaxosmithkline-300591034.html
- Ramadon, D., McCrudden, M. T. C., Courtenay, A. J., Donnelly, R. F. (2021). Enhancement strategies for transdermal drug delivery systems: current trends and applications. Drug Delivery and Translational Research, 12 (4), 758–791. https://doi.org/10.1007/s13346-021-00909-6
- Uttekar, P., Kulkarni, A., Chaudhari, P., Dhage, M., Dhangarmali, V. (2016). Formulation and evaluation of captopril transdermal patches for the treatment of hypertension. Der Pharmacia Lettre, 8 (5). 12–16. Available at: https://www.scholarsresearchlibrary.com/articles/formulation-and-evaluation-of-captopril-transdermal-patches-for-thetreatment-of-hypertension.pdf
- Debasmita, G., Bhowmick, M., Bhowmick, P. (2023). Fabrication and characterization of ace inhibitor loaded transdermal patches. International Journal of Allied Medical Sciences and Clinical Research, 11 (2), 152–161.
- Ramadon, D., Muliawardani, F., Nisrina, N. A., Tri Hamda, O., Iswandana, R., Wahyuni, T. et al. (2024). Transdermal delivery of captopril using poly(vinyl pyrrolidone)/poly(vinyl alcohol)-based dissolving and hydrogel-forming microneedles: A proof of concept. European Polymer Journal, 208, 112860. https://doi.org/10.1016/j.eurpolymj.2024.112860
- Shyteyeva, T., Bezchasnyuk, E., Kryskiv, O. (2022) In vitro Study of the Permeability of Enalapril Maleate through a Semipermeable Membrane in the Process of Pharmaceutical Development of a Transdermal Therapeutic System. Pharmakeftiki, 34 (4), 166–173. Available at: https://pharmakeftiki.hsmc.gr/pj/issue/view/22/22
- Shyteyeva, T., Bezchasnyuk, E., Kryskiv, O., Baranova, I. (2024). Biopharmaceutical aspects of the development of transdermal forms of Lisinopril dihydrate. Current Issues in Pharmacy and Medical Sciences, 37 (3), 166–170. https://doi.org/10.2478/cipms-2024-0027
- Babiker, M. E., Farah, H. F., Heyam, S. A. (2021) Captopril: An Overview of Discovery, Develop and Post-marketing Surveillan ceasan Effective Anti-hypertensive Drug. Acta Scientific Pharmaceutical Sciences, 5 (4). 6–9. Available at: https://actascientific.com/ASPS/ASPS-05-0695.php
- Chien, Y. W. (1987). Development of Transdermal Drug Delivery Systems. Drug Development and Industrial Pharmacy, 13 (4-5), 589–651. https://doi.org/10.3109/03639048709105212
- Kaptopryl (2014) Derzhavna farmakopeia Ukrainy. Vol. 2. Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 347–350.
- Captopril (Captoprilum) (2016). The International Pharmacopoeia. Available at: https://cdn.who.int/media/docs/default-source/medicines/pharmacopoeia/omitted-monographs/captopril.pdf?sfvrsn=7d2ac6d4_5
- 3.N.1. Statystychnyi analiz rezultativ khimichnoho eksperymentu (2023) Derzhavna farmakopeia Ukrainy (druhe vydannia). Dopovnennia 6. Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 30–40.
- 9.6. Uniformity of content of single-dose preparations (2016) Ph. Eur. 10.0. Strasbourg, France: Council of Europe, 336. Available at: https://www.scribd.com/document/508063535/European-Pharmacopoeia-10-0#page=368
##submission.downloads##
Опубліковано
Як цитувати
Номер
Розділ
Ліцензія
Авторське право (c) 2026 Tatyana Shyteyeva, Elena Bezchasnyuk, Oleg Kryskiv

Ця робота ліцензується відповідно до Creative Commons Attribution 4.0 International License.
Наше видання використовує положення про авторські права Creative Commons Attribution 4.0 International License для журналів відкритого доступу.
Автори, які публікуються у цьому журналі, погоджуються з наступними умовами:
1. Автори залишають за собою право на авторство своєї роботи та передають журналу право першої публікації цієї роботи на умовах ліцензії Creative Commons Attribution 4.0 International License, котра дозволяє іншим особам вільно розповсюджувати опубліковану роботу з обов'язковим посиланням на авторів оригінальної роботи та першу публікацію роботи у цьому журналі.
2. Автори мають право укладати самостійні додаткові угоди щодо неексклюзивного розповсюдження роботи у тому вигляді, в якому вона була опублікована цим журналом (наприклад, розміщувати роботу в електронному сховищі установи або публікувати у складі монографії), за умови збереження посилання на першу публікацію роботи у цьому журналі.
3. Автори мають право зберігати остаточну прийняту версію статті в інституційному, тематичному або будь-якому іншому репозитарії з метою забезпечення видимості та доступності.







