Determination of the possibility of obtaining ballastless Ni(II)-Ni(III) layered double hydroxides as promising active substances for supercapacitors and electrocatalysis
DOI:
https://doi.org/10.15587/1729-4061.2026.352268Keywords:
coprecipitation at high supersaturation, coprecipitation at constant pH, Ni2 -Ni3 LDH, sodium hypochloriteAbstract
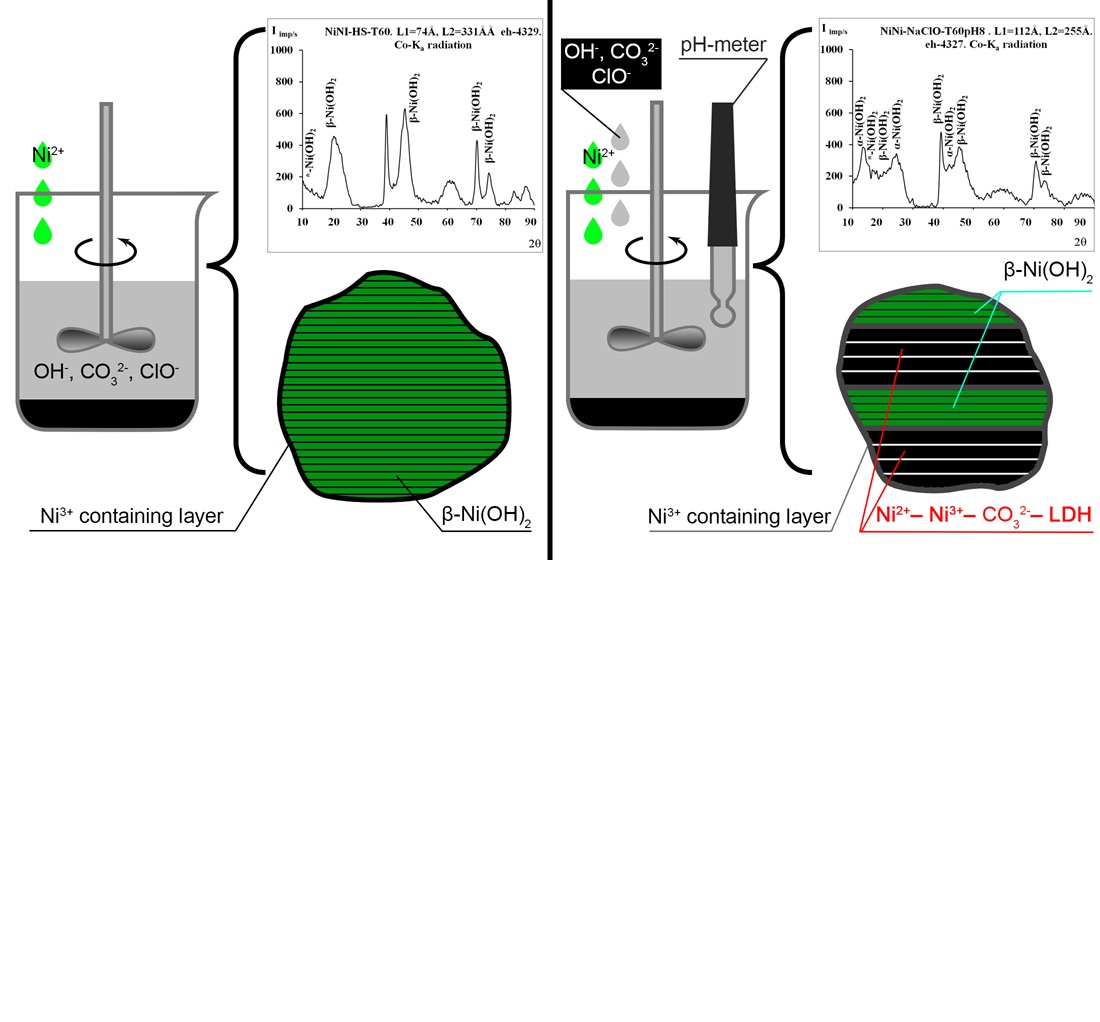
This study is aimed to investigate the synthesis of Ni2⁺-Ni3⁺ LDH (layered double hydroxide) in the presence of sodium hypochlorite. Ni2⁺-Ni3⁺ LDH is a promising active material for use in supercapacitors and electrocatalysis. The syntheses were carried out at a temperature of 60°C by coprecipitation under conditions of high supersaturation and at constant pH values (8, 10, and 12). To obtain the guest metal cation Ni3⁺ from the initial Ni2⁺ during synthesis, sodium hypochlorite was introduced as an oxidizing agent into the alkaline sodium carbonate solution. As control samples, Ni-Al-carbonate LDH were synthesized using the same methods and conditions. The formation of Ni3⁺ hydroxo compounds during synthesis was visually confirmed by a color change. The crystal structure of the samples was investigated by X-ray diffraction analysis, and the total Ni and Ni3⁺ contents were determined by trilonometric and iodometric titration. The samples synthesized in the presence of hypochlorite exhibited a black color, confirming the successful formation of Ni3⁺ hydroxo compounds. All control samples corresponded to Ni-Al LDH. The nickel hydroxide sample synthesized by coprecipitation at high supersaturation was identified as β-Ni(OH)2, with a total Ni content of 59.5% and a Ni3⁺ fraction of 12.2%. The transformation of Ni2⁺ → Ni3⁺ occurred in the surface layer of the formed β-Ni(OH)2 particles because the oxidation rate was lower than the hydroxide formation rate. The samples synthesized by coprecipitation at constant pH exhibited a layered structure consisting of β-Ni(OH)2 and Ni2⁺–Ni3⁺ LDH with an α-type lattice, suggesting that the oxidation rate exceeded the hydroxide formation rate. Thus, the possibility of synthesizing Ni2⁺-Ni3⁺-carbonate LDH was experimentally demonstrated. Under the optimal conditions (pH 8), the proportion of the Ni-Ni LDH phase reached 55.9%. The synthesized Ni2⁺-Ni3⁺ LDH shows potential for application in supercapacitors and electrocatalysis, provided that its specific electrochemical characteristics are determined in further studies
References
- Kotok, V., Kovalenko, V. (2018). Definition of the aging process parameters for nickel hydroxide in the alkaline medium. Eastern-European Journal of Enterprise Technologies, 2 (12 (92)), 54–60. https://doi.org/10.15587/1729-4061.2018.127764
- Liang, H., Lin, J., Jia, H., Chen, S., Qi, J., Cao, J., Lin, T., Fei, W., Feng, J. (2018). Hierarchical NiCo-LDH/NiCoP@NiMn-LDH hybrid electrodes on carbon cloth for excellent supercapacitors. Journal of Materials Chemistry A, 6 (31), 15040–15046. https://doi.org/10.1039/c8ta05065a
- Kovalenko, V., Kotok, V., Kovalenko, I. (2018). Activation of the nickel foam as a current collector for application in supercapacitors. Eastern-European Journal of Enterprise Technologies, 3 (12 (93)), 56–62. https://doi.org/10.15587/1729-4061.2018.133472
- Kotok, V. A., Kovalenko, V. L., Solovov, V. A., Kovalenko, P. V., Ananchenko, B. A. (2018). Effect of deposition time on properties of electrochromic nickel hydroxide films prepared by cathodic template synthesis. ARPN Journal of Engineering and Applied Sciences, 13 (9), 3076–3086. Available at: https://www.arpnjournals.org/jeas/research_papers/rp_2018/jeas_0518_7034.pdf
- Wang, Y., Zhang, D., Peng, W., Liu, L., Li, M. (2011). Electrocatalytic oxidation of methanol at Ni–Al layered double hydroxide film modified electrode in alkaline medium. Electrochimica Acta, 56 (16), 5754–5758. https://doi.org/10.1016/j.electacta.2011.04.049
- Solovov, V. A., Nikolenko, N. V., Kovalenko, V. L., Kotok, V. A., Burkov, A. А., Kondrat’ev, D. A. et. al. (2018). Synthesis of Ni(II)-Ti(IV) Layered Double Hydroxides Using Coprecipitation At High Supersaturation Method. ARPN Journal of Engineering and Applied Sciences, 24 (13), 9652–9656. Avaiable at: https://www.arpnjournals.org/jeas/research_papers/rp_2018/jeas_1218_7500.pdf
- Zhao, Y., Wei, M., Lu, J., Wang, Z. L., Duan, X. (2009). Biotemplated Hierarchical Nanostructure of Layered Double Hydroxides with Improved Photocatalysis Performance. ACS Nano, 3 (12), 4009–4016. https://doi.org/10.1021/nn901055d
- Su, Q., Gu, L., Yao, Y., Zhao, J., Ji, W., Ding, W., Au, C.-T. (2017). Layered double hydroxides derived Nix(MgyAlzOn) catalysts: Enhanced ammonia decomposition by hydrogen spillover effect. Applied Cриatalysis B: Environmental, 201, 451–460. https://doi.org/10.1016/j.apcatb.2016.08.051
- Fedorova, Z. A., Borisov, V. A., Pakharukova, V. P., Gerasimov, E. Y., Belyaev, V. D., Gulyaeva, T. I. et al. (2023). Layered Double Hydroxide-Derived Ni-Mg-Al Catalysts for Ammonia Decomposition Process: Synthesis and Characterization. Catalysts, 13 (4), 678. https://doi.org/10.3390/catal13040678
- Fan, Y., Yang, Z., Cao, X., Liu, P., Chen, S., Cao, Z. (2014). Hierarchical Macro-Mesoporous Ni(OH)2for Nonenzymatic Electrochemical Sensing of Glucose. Journal of The Electrochemical Society, 161 (10), B201–B206. https://doi.org/10.1149/2.0251410jes
- Ramesh, T. N., Kamath, P. V. (2006). Synthesis of nickel hydroxide: Effect of precipitation conditions on phase selectivity and structural disorder. Journal of Power Sources, 156 (2), 655–661. https://doi.org/10.1016/j.jpowsour.2005.05.050
- Rajamathi, M., Vishnu Kamath, P., Seshadri, R. (2000). Polymorphism in nickel hydroxide: role of interstratification. Journal of Materials Chemistry, 10 (2), 503–506. https://doi.org/10.1039/a905651c
- Kovalenko, V., Kotok, V. (2019). Influence of the carbonate ion on characteristics of electrochemically synthesized layered (α+β) nickel hydroxide. Eastern-European Journal of Enterprise Technologies, 1 (6 (97)), 40–46. https://doi.org/10.15587/1729-4061.2019.155738
- Jayashree, R. S., Vishnu Kamath, P. (2001). Suppression of the α → β-nickel hydroxide transformation in concentrated alkali: Role of dissolved cations. Journal of Applied Electrochemistry, 31 (12), 131–1320. https://doi.org/10.1023/a:1013876006707
- Córdoba de Torresi, S. I., Provazi, K., Malta, M., Torresi, R. M. (2001). Effect of Additives in the Stabilization of the α Phase of Ni(OH)2 Electrodes. Journal of The Electrochemical Society, 148 (10), A1179–A1184. https://doi.org/10.1149/1.1403731
- Kovalenko, V., Kotok, V., Yeroshkina, A., Zaychuk, A. (2017). Synthesis and characterisation of dyeintercalated nickelaluminium layereddouble hydroxide as a cosmetic pigment. Eastern-European Journal of Enterprise Technologies, 5 (12 (89)), 27–33. https://doi.org/10.15587/1729-4061.2017.109814
- Shivaramaiah, R., Navrotsky, A. (2015). Energetics of Order–Disorder in Layered Magnesium Aluminum Double Hydroxides with Interlayer Carbonate. Inorganic Chemistry, 54 (7), 3253–3259. https://doi.org/10.1021/ic502820q
- Kovalenko, V., Kotok, V. (2020). Bifuctional indigocarminintercalated NiAl layered double hydroxide: investigation of characteristics for pigment and supercapacitor application. Eastern-European Journal of Enterprise Technologies, 2 (12 (104)), 30–39. https://doi.org/10.15587/1729-4061.2020.201282
- Li, Y. W., Yao, J. H., Liu, C. J., Zhao, W. M., Deng, W. X., Zhong, S. K. (2010). Effect of interlayer anions on the electrochemical performance of Al-substituted α-type nickel hydroxide electrodes. International Journal of Hydrogen Energy, 35(6), 2539–2545. https://doi.org/10.1016/j.ijhydene.2010.01.015
- Qi, J., Xu, P., Lv, Z., Liu, X., Wen, A. (2008). Effect of crystallinity on the electrochemical performance of nanometer Al-stabilized α-nickel hydroxide. Journal of Alloys and Compounds, 462 (1-2), 164–169. https://doi.org/10.1016/j.jallcom.2007.07.102
- Li, H., Chen, Z., Wang, Y., Zhang, J., Yan, X. (2016). Controlled synthesis and enhanced electrochemical performance of self-assembled rosette-type Ni-Al layered double hydroxide. Electrochimica Acta, 210, 15–22. https://doi.org/10.1016/j.electacta.2016.05.132
- Bao, J., Zhu, Y. J., Xu, Q. S., Zhuang, Y. H., Zhao, R. D., Zeng, Y. Y., Zhong, H. L. (2012). Structure and Electrochemical Performance of Cu and Al Codoped Nanometer α-Nickel Hydroxide. Advanced Materials Research, 479-481, 230–233. https://doi.org/10.4028/www.scientific.net/amr.479-481.230
- Kovalenko, V. L., Kotok, V. A., Sykchin, A., Ananchenko, B. A., Chernyad’ev, A. V., Burkov, A. A. et al. (2020). Al3+ Additive in the Nickel Hydroxide Obtained by High-Temperature Two-Step Synthesis: Activator or Poisoner for Chemical Power Source Application? Journal of The Electrochemical Society, 167 (10), 100530. https://doi.org/10.1149/1945-7111/ab9a2a
- Huang, J., Lei, T., Wei, X., Liu, X., Liu, T., Cao, D., Yin, J., Wang, G. (2013). Effect of Al-doped β-Ni(OH)2 nanosheets on electrochemical behaviors for high performance supercapacitor application. Journal of Power Sources, 232, 370–375. https://doi.org/10.1016/j.jpowsour.2013.01.081
- Memon, J., Sun, J., Meng, D., Ouyang, W., Memon, M. A., Huang, Y. et al. (2014). Synthesis of graphene/Ni–Al layered double hydroxide nanowires and their application as an electrode material for supercapacitors. Journal of Materials Chemistry A, 2 (14), 5060–5067. https://doi.org/10.1039/c3ta14613h
- Mignani, A., Ballarin, B., Giorgetti, M., Scavetta, E., Tonelli, D., Boanini, E. et al. (2013). Heterostructure of Au Nanoparticles – NiAl Layered Double Hydroxide: Electrosynthesis, Characterization, and Electrocatalytic Properties. The Journal of Physical Chemistry C, 117 (31), 16221–16230. https://doi.org/10.1021/jp4033782
- Kovalenko, V., Kotok, V. (2019). Investigation of characteristics of double Ni–Co and ternary Ni–Co–Al layered hydroxides for supercapacitor application. Eastern-European Journal of Enterprise Technologies, 2 (6 (98)), 58–66. https://doi.org/10.15587/1729-4061.2019.164792
- Kovalenko, V., Kotok, V. (2021). The determination of synthesis conditions and color properties of pigments based on layered double hydroxides with Co as a guest cation. Eastern-European Journal of Enterprise Technologies, 6 (6 (114)), 32–38. https://doi.org/10.15587/1729-4061.2021.247160
- Radha, A. V., Kamath, P. V. (2004). Oxidative leaching of chromium from layered double hydroxides: Mechanistic studies. Bulletin of Materials Science, 27 (4), 355–360. https://doi.org/10.1007/bf02704772
- Xu, L., Wang, Z., Chen, X., Qu, Z., Li, F., Yang, W. (2018). Ultrathin layered double hydroxide nanosheets with Ni(III) active species obtained by exfoliation for highly efficient ethanol electrooxidation. Electrochimica Acta, 260, 898–904. https://doi.org/10.1016/j.electacta.2017.12.065
- Nestroinaia, O. V., Ryltsova, I. G., Lebedeva, O. E. (2021). Effect of Synthesis Method on Properties of Layered Double Hydroxides Containing Ni(III). Crystals, 11 (11), 1429. https://doi.org/10.3390/cryst11111429
- Ryltsova, I. G., Nestroinaya, O. V., Lebedeva, O. E., Schroeter, F., Roessner, F. (2018). Synthesis and characterization of layered double hydroxides containing Nickel in unstable oxidation state + 3 in cationic sites. Journal of Solid State Chemistry, 265, 332–338. https://doi.org/10.1016/j.jssc.2018.06.001
- Ryltsova, I. G., Nestroinaia, O. V., Lebedeva, O. E. (2019). Nickel(III) containing layered double hydroxides with hydrotalcite structure. Butlerov Communications, 57 (2), 116–122. https://doi.org/10.37952/roi-jbc-01/19-57-2-116
- Kovalenko, V., Kotok, V. (2019). Anionic carbonate activation of layered (α+β) nickel hydroxide. Eastern-European Journal of Enterprise Technologies, 3(6 (99)), 44–52. https://doi.org/10.15587/1729-4061.2019.169461
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Vadym Kovalenko, Dmytro Andreiev, Valerii Kotok, Alexander Baskevich, Volodymyr Medianyk, Dmytro Sukhomlyn, Volodymyr Verbitskiy

This work is licensed under a Creative Commons Attribution 4.0 International License.
The consolidation and conditions for the transfer of copyright (identification of authorship) is carried out in the License Agreement. In particular, the authors reserve the right to the authorship of their manuscript and transfer the first publication of this work to the journal under the terms of the Creative Commons CC BY license. At the same time, they have the right to conclude on their own additional agreements concerning the non-exclusive distribution of the work in the form in which it was published by this journal, but provided that the link to the first publication of the article in this journal is preserved.
A license agreement is a document in which the author warrants that he/she owns all copyright for the work (manuscript, article, etc.).
The authors, signing the License Agreement with TECHNOLOGY CENTER PC, have all rights to the further use of their work, provided that they link to our edition in which the work was published.
According to the terms of the License Agreement, the Publisher TECHNOLOGY CENTER PC does not take away your copyrights and receives permission from the authors to use and dissemination of the publication through the world's scientific resources (own electronic resources, scientometric databases, repositories, libraries, etc.).
In the absence of a signed License Agreement or in the absence of this agreement of identifiers allowing to identify the identity of the author, the editors have no right to work with the manuscript.
It is important to remember that there is another type of agreement between authors and publishers – when copyright is transferred from the authors to the publisher. In this case, the authors lose ownership of their work and may not use it in any way.





