Engineering chitosan-Exenatide nanocomplexes with fatty acids for improved peptide delivery
DOI:
https://doi.org/10.15587/2519-4852.2026.340895Keywords:
Exenatide, chitosan, fatty acids, polyelectrolyte complex (PEC)Abstract
Diabetes Mellitus (DM) is a common endocrine disorder responsible for high morbidity and mortality worldwide. The standard medical treatment for DM is oral hypoglycemic agents and/or insulin. Exenatide, a glucagon-like peptide, has been used to reduce blood sugar and treat DM in the last 20 years. Exenatide administration is limited to parenteral routes. The development of an orally administered Exenatide represents a worthy study that provides significant benefits to patients with diabetes by improving compliance and adherence to the treatment and reduce the burden of frequent injections and enhance treatment outcomes.
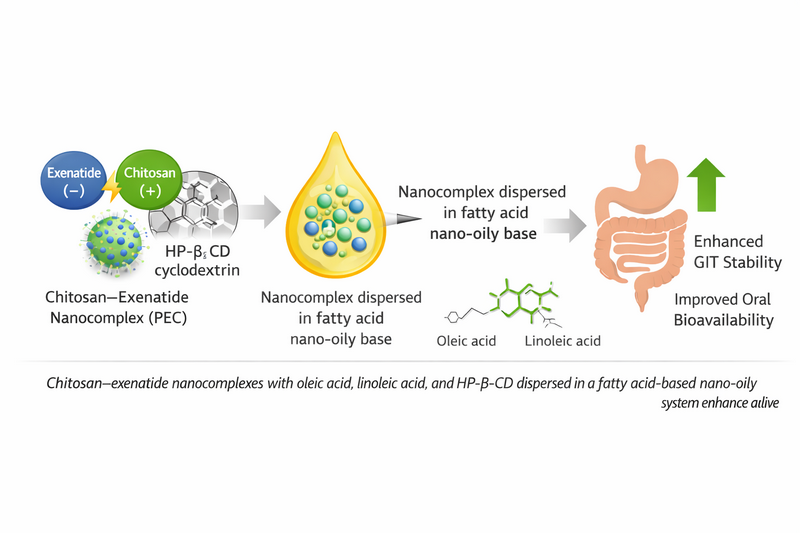
The aim of the study is to prepare Exenatide as an oral drug delivery system by combining the advantages of nanoencapsulation with the use of an oily vehicle using fatty acids.
Method: The polyelectrolyte complexation method was used to prepare Exenatide-chitosan complexes (PEC) as an aqueous environment in order to create orally administered Exenatide. The potential of PEC -fatty acids nanoparticles as oral delivery carriers of Exenatide was studied.
Results: The sizes of the formed nanodispersed particles were different when loaded with diluted chitosan or PEC. The vortex mechanical mixing method produced superior results and provided about 20% greater Exenatide gastrointestinal protection than the stirring mechanical method. The results indicated that hydroxypropyl-β-cyclodextrin (HP-β-CD) had a more promising effect on oleic acid formula (F4), providing 87.1% Exenatide gastrointestinal protection but with a larger nanodispersed particle size of 200 nm. However, it did not produce significantly better results for linoleic acid (F8), which provided 81.6% gastrointestinal protection and a nanodispersed particle size of 210 nm. An in vivo study showed that formula F4 has the Cmax of Exenatide with Tmax of 3 h. Blood glucose was effectively reduced to a level of 91 mg/dl level within 3 h, with a sustained reduction up to 8 h.
Conclusion: Exenatide could be protected from gastrointestinal enzymes by incorporation into chitosan lipid-based formulation. The vortexing mechanical mixing method is preferred method for the preparation. The use of the HP-β-CD improved gastrointestinal protection. The formula F4 is a promising oral alternative to the paraenteral Exenatide
References
- Shi, Y., Sun, X., Zhang, L., Sun, K., Li, K., Li, Y., Zhang, Q. (2018). Fc-modified exenatide-loaded nanoparticles for oral delivery to improve hypoglycemic effects in mice. Scientific Reports, 8 (1). https://doi.org/10.1038/s41598-018-19170-y
- Yang, J.-M., Wu, L.-J., Lin, M.-T., Lu, Y.-Y., Wang, T.-T., Han, M. et al. (2022). Construction and Evaluation of Chitosan-Based Nanoparticles for Oral Administration of Exenatide in Type 2 Diabetic Rats. Polymers, 14 (11), 2181. https://doi.org/10.3390/polym14112181
- Phan, T. N. Q., Ismail, R., Le-Vinh, B., Zaichik, S., Laffleur, F., Bernkop-Schnürch, A. (2020). The Effect of Counterions in Hydrophobic Ion Pairs on Oral Bioavailability of Exenatide. ACS Biomaterials Science & Engineering, 6 (9), 5032–5039. https://doi.org/10.1021/acsbiomaterials.0c00637
- Aranaz, I., Alcántara, A. R., Civera, M. C., Arias, C., Elorza, B., Heras Caballero, A., Acosta, N. (2021). Chitosan: An Overview of Its Properties and Applications. Polymers, 13 (19), 3256. https://doi.org/10.3390/polym13193256
- Sweidan, K., Jaber, A.-M., Al-Jbour, N., Obaidat, R., Al Remawi, M., Badwan, A. (2011). Further investigation on the degree of deacetylation of chitosan determined by potentiometric titration. Journal of Excipients and Food Chemistry, 2 (1), 16–25.
- Naso, J. N., Bellesi, F. A., Pizones Ruiz-Henestrosa, V. M., Pilosof, A. M. R. (2021). A new methodology to assess the solubility of fatty acids: Impact of food emulsifiers. Food Research International, 139, 109829. https://doi.org/10.1016/j.foodres.2020.109829
- Elsayed, A., Remawi, M. A., Qinna, N., Farouk, A., Badwan, A. (2009). Formulation and characterization of an oily-based system for oral delivery of insulin. European Journal of Pharmaceutics and Biopharmaceutics, 73 (2), 269–279. https://doi.org/10.1016/j.ejpb.2009.06.004
- Ismail, R., Phan, T. N. Q., Laffleur, F., Csóka, I., Bernkop-Schnürch, A. (2020). Hydrophobic ion pairing of a GLP-1 analogue for incorporating into lipid nanocarriers designed for oral delivery. European Journal of Pharmaceutics and Biopharmaceutics, 152, 10–17. https://doi.org/10.1016/j.ejpb.2020.04.025
- Douglas-Gallardo, O. A., Christensen, C. A., Strumia, M. C., Pérez, M. A., Gomez, C. G. (2019). Physico-chemistry of a successful micro-reactor: Random coils of chitosan backbones used to synthesize size-controlled silver nanoparticles. Carbohydrate Polymers, 225, 115241. https://doi.org/10.1016/j.carbpol.2019.115241
- Younes, I., Rinaudo, M. (2015). Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Marine Drugs, 13 (3), 1133–1174. https://doi.org/10.3390/md13031133
- Yuan, H., Xiao, P., Wang, F., Guo, C., Pan, S., Jiang, M. et al. (2025). Linoleic acid co-administration promotes oral delivery of exenatide-loaded butyrate-decorated nanocapsules. Journal of Controlled Release, 382, 113744. https://doi.org/10.1016/j.jconrel.2025.113744
- Tekeli, M. C., Aktas, Y., Celebi, N. (2021). Oral self-nanoemulsifying formulation of GLP-1 agonist peptide exendin-4: development, characterization and permeability assesment on Caco-2 cell monolayer. Amino Acids, 53 (1), 73–88. https://doi.org/10.1007/s00726-020-02926-0
- Zulfakar, M. H., Pubadi, H., Ibrahim, S. I., Hairul, N. M. (2024). Medium-Chain Triacylglycerols (MCTs) and Their Fractions in Drug Delivery Systems : A Systematic Review. Journal of Oleo Science, 73 (3), 293–310. https://doi.org/10.5650/jos.ess23204
- Smail, S. S., Ghareeb, M. M., Omer, H. K., Al-Kinani, A. A., Alany, R. G. (2021). Studies on Surfactants, Cosurfactants, and Oils for Prospective Use in Formulation of Ketorolac Tromethamine Ophthalmic Nanoemulsions. Pharmaceutics, 13 (4), 467. https://doi.org/10.3390/pharmaceutics13040467
- Djekic, L., Primorac, M. (2008). The influence of cosurfactants and oils on the formation of pharmaceutical microemulsions based on PEG-8 caprylic/capric glycerides. International Journal of Pharmaceutics, 352 (1-2), 231–239. https://doi.org/10.1016/j.ijpharm.2007.10.041
- Ibrahim, S. A., Li, S. K. (2009). Efficiency of Fatty Acids as Chemical Penetration Enhancers: Mechanisms and Structure Enhancement Relationship. Pharmaceutical Research, 27 (1), 115–125. https://doi.org/10.1007/s11095-009-9985-0
- Mukherjee, S., Shanmugam, G. (2023). A Novel Surfactant with Short Hydrophobic Head and Long Hydrophilic Tail Generates Vesicles with Unique Structural Feature. Small, 19 (19). https://doi.org/10.1002/smll.202206906
- Ribeiro, E. F., de Barros-Alexandrino, T. T., Assis, O. B. G., Junior, A. C., Quiles, A., Hernando, I., Nicoletti, V. R. (2020). Chitosan and crosslinked chitosan nanoparticles: Synthesis, characterization and their role as Pickering emulsifiers. Carbohydrate Polymers, 250, 116878. https://doi.org/10.1016/j.carbpol.2020.116878
- Benamer Oudih, S., Tahtat, D., Nacer Khodja, A., Mahlous, M., Hammache, Y., Guittoum, A., Kebbouche Gana, S. (2023). Chitosan nanoparticles with controlled size and zeta potential. Polymer Engineering & Science, 63 (3), 1011–1021. https://doi.org/10.1002/pen.26261
- Butnaru, E., Stoleru, E., Brebu, M. A., Darie-Nita, R. N., Bargan, A., Vasile, C. (2019). Chitosan-Based Bionanocomposite Films Prepared by Emulsion Technique for Food Preservation. Materials, 12 (3), 373. https://doi.org/10.3390/ma12030373
- Vargas, M., Albors, A., Chiralt, A., González-Martínez, C. (2009). Characterization of chitosan–oleic acid composite films. Food Hydrocolloids, 23 (2), 536–547. https://doi.org/10.1016/j.foodhyd.2008.02.009
- Brunel, F., Véron, L., David, L., Domard, A., Delair, T. (2008). A Novel Synthesis of Chitosan Nanoparticles in Reverse Emulsion. Langmuir, 24 (20), 11370–11377. https://doi.org/10.1021/la801917a
- Eczacioglu, N., Postina, A., Ebert, M., Laffleur, F., Kali, G., Seybold, A., Bernkop-Schnürch, A. (2025). Self-emulsifying drug delivery systems: A comparison of dry and wet reverse micelles. Acta Biomaterialia, 202, 545–558. https://doi.org/10.1016/j.actbio.2025.07.027
- Álvarez-García, S., Couarraze, L., Matos, M., Gutiérrez, G. (2024). Lycopene-Loaded Emulsions: Chitosan Versus Non-Ionic Surfactants as Stabilizers. Molecules, 29 (21), 5209. https://doi.org/10.3390/molecules29215209
- Bowman, K., Leong, K. W. (2006). Chitosan nanoparticles for oral drug and gene delivery. International Journal of Nanomedicine, 1 (2), 117–128. https://doi.org/10.2147/nano.2006.1.2.117
- Claus, V., Spleis, H., Federer, C., Zöller, K., Wibel, R., Laffleur, F. et al. (2023). Self-emulsifying drug delivery systems (SEDDS): In vivo-proof of concept for oral delivery of insulin glargine. International Journal of Pharmaceutics, 639, 122964. https://doi.org/10.1016/j.ijpharm.2023.122964
- Li, H., Zhang, Z., Bao, X., Xu, G., Yao, P. (2018). Fatty acid and quaternary ammonium modified chitosan nanoparticles for insulin delivery. Colloids and Surfaces B: Biointerfaces, 170, 136–143. https://doi.org/10.1016/j.colsurfb.2018.05.063
- Kuroiwa, T., Shino, H., Yoshioka, T., Doi, T., Nishinomiya, T. (2022). Flavor encapsulation into chitosan-oleic acid complex particles and its controlled release characteristics during heating processes. LWT, 167, 113815. https://doi.org/10.1016/j.lwt.2022.113815
- Kurniawan, J., Suga, K., Kuhl, T. L. (2017). Interaction forces and membrane charge tunability: Oleic acid containing membranes in different pH conditions. Biochimica et Biophysica Acta (BBA) – Biomembranes, 1859 (2), 211–217. https://doi.org/10.1016/j.bbamem.2016.11.001
- Boughanmi, R., Oelmann, M., Steinbach, C., Schwarz, S. (2024). Comparative Study on Polyelectrolyte Complex Formation of Chitosan and Pectin or PEMA: Effects of Molecular Weight and Mixing Speed. Polysaccharides, 5 (4), 842–856. https://doi.org/10.3390/polysaccharides5040052
- Tiwari, G., Tiwari, R., Rai, A. (2010). Cyclodextrins in delivery systems: Applications. Journal of Pharmacy And Bioallied Sciences, 2 (2), 72–79. https://doi.org/10.4103/0975-7406.67003
- Zapadka, K. L., Becher, F. J., Gomes dos Santos, A. L., Jackson, S. E. (2017). Factors affecting the physical stability (aggregation) of peptide therapeutics. Interface Focus, 7 (6), 20170030. https://doi.org/10.1098/rsfs.2017.0030
- Musuc, A. M. (2024). Cyclodextrins: Advances in Chemistry, Toxicology, and Multifaceted Applications. Molecules, 29 (22), 5319. https://doi.org/10.3390/molecules29225319
- Kali, G., Haddadzadegan, S., Bernkop-Schnürch, A. (2024). Cyclodextrins and derivatives in drug delivery: New developments, relevant clinical trials, and advanced products. Carbohydrate Polymers, 324, 121500. https://doi.org/10.1016/j.carbpol.2023.121500
- Sarabia-Vallejo, Á., Caja, M. del M., Olives, A. I., Martín, M. A., Menéndez, J. C. (2023). Cyclodextrin Inclusion Complexes for Improved Drug Bioavailability and Activity: Synthetic and Analytical Aspects. Pharmaceutics, 15 (9), 2345. https://doi.org/10.3390/pharmaceutics15092345
- Cid-Samamed, A., Rakmai, J., Mejuto, J. C., Simal-Gandara, J., Astray, G. (2022). Cyclodextrins inclusion complex: Preparation methods, analytical techniques and food industry applications. Food Chemistry, 384, 132467. https://doi.org/10.1016/j.foodchem.2022.132467
- Fonte, P., Nogueira, T., Gehm, C., Ferreira, D., Sarmento, B. (2011). Chitosan-coated solid lipid nanoparticles enhance the oral absorption of insulin. Drug Delivery and Translational Research, 1 (4), 299–308. https://doi.org/10.1007/s13346-011-0023-5
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Rana Hani Mohammed Ali Al-Shaikh Hamed, Muhammed Hameed Faeadh Hsn Al-Jumaily

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






