Development of two spectrophotometric methods for the determination of bilastine in tablets
DOI:
https://doi.org/10.15587/2519-4852.2026.352766Keywords:
bilastine, tablets, spectrophotometry, sulfophthalein dyes, validation, quantitative determinationAbstract
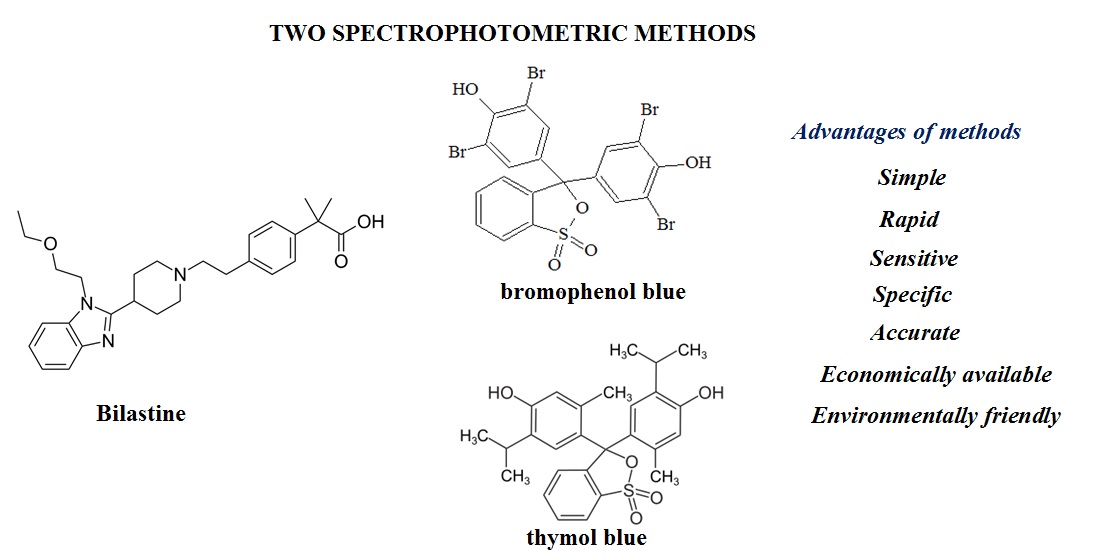
The aim of the work was to develop two simple, rapid, economically available spectrophotometric methods for the determination of bilastine in tablets based on the reaction with sulfonephthalein dyes (bromphenol blue (BPB) and thymol blue (TB)).
Materials and methods. Analytical instrumentation: Shimadzu UV-1800 double beam UV-VIS spectrophotometer (Japan) with attached UV-Probe ver. 2.62 software, RAD WAG AS 200/C precise analytical balance (Poland). Bilastine (purity ≥99% (LC)) was purchased from Ukrainian Scientific Pharmacopoeial Center for Quality of Medicines. Nixar tablets 20 mg were purchased from a local pharmacy.
Results and discussion. Two spectrophotometric methods for the determination of bilastine in tablets have been developed. Different sulfophthalein dyes (bromphenol blue, thymol blue, bromocresol green, bromthymol blue, bromocresol purple) have been tested in order to choose the optimal reagent for the method development. The experimental research results led to the selection of BPB and TB as the reagents. Methanol was used as the solvent in reaction of bilastine with BPB, while 20% methanol-ethyl acetate solution was used for TB.
The optimal conditions for the quantitative determination of bilastine in tablets by using BPB were established: concentration – 1.08×10-3 mol/L, volume of BPB solution – 1.00 mL, wavelength – 596 nm, reaction time – 5 min, solution temperature – 25°C. The optimal conditions for the quantitative determination of bilastine in tablets by using TB were established: concentration – 4.34×10-4 mol/L, volume of TB solution – 1.00 mL, wavelength – 416 nm, reaction time – 5 min, solution temperature – 25°C.
The spectrophotometric method of the quantitative determination of bilastine in tablets by using BPB was linear in the concentration range of 0.5-7.5 μg/mL, LOD – 0.25 μg/mL, LOQ – 0.76 μg/mL; by using TB was linear in the concentration range of 2.00–18.00 μg/mL, LOD – 0.63 μg/mL, LOQ – 1.92 μg/mL. Both methods demonstrated acceptable robustness, accuracy, and precision, meeting all validation criteria. The «greenness» assessment results confirmed that both methods are excellent from a green analytical chemistry perspective.
Conclusions. The developed methods can be used as an alternative method for the routine analysis of bilastine in tablets
References
- Wang, J., Zhou, Y., Zhang, H., Hu, L., Liu, J., Wang, L. et al. (2023). Pathogenesis of allergic diseases and implications for therapeutic interventions. Signal Transduction and Targeted Therapy, 8 (1). https://doi.org/10.1038/s41392-023-01344-4
- Varshney, J., Varshney, H. (2015). Allergic Rhinitis: an Overview. Indian Journal of Otolaryngology and Head & Neck Surgery, 67 (2), 143–149. https://doi.org/10.1007/s12070-015-0828-5
- Parisi, G. F., Licari, A., Papale, M., Manti, S., Salpietro, C., Marseglia, G. L., Leonardi, S. (2020). Antihistamines: ABC for the pediatricians. Pediatric Allergy and Immunology, 31 (S24), 34–36. Portico. https://doi.org/10.1111/pai.13152
- Kawauchi, H., Yanai, K., Wang, D.-Y., Itahashi, K., Okubo, K. (2019). Antihistamines for Allergic Rhinitis Treatment from the Viewpoint of Nonsedative Properties. International Journal of Molecular Sciences, 20 (1), 213. https://doi.org/10.3390/ijms20010213
- Bilastine. Available at: https://pubchem.ncbi.nlm.nih.gov/compound/185460 Last accessed: 01.05.2025
- Church, M. K., Tiongco-Recto, M., Ridolo, E., Novák, Z. (2019). Bilastine: a lifetime companion for the treatment of allergies. Current Medical Research and Opinion, 36 (3), 445–454. https://doi.org/10.1080/03007995.2019.1681134
- Demonte, A., Guanti, M. B., Liberati, S., Biffi, A., Fernando, F., Fainello, M., Pepe, P. (2018). Bilastine safety in drivers who need antihistamines: new evidence from high-speed simulator driving test on allergic patients. European review for medical and pharmacological sciences, 22 (3), 820–828. https://doi.org/10.26355/eurrev_201802_14318
- Kowal, K., DuBuske, L. (2014). Bilastine as a Potential Treatment in Allergic Rhinitis. American Journal of Rhinology & Allergy, 28 (4), 312–316. https://doi.org/10.2500/ajra.2014.28.4049
- Bilastine tablets (monograph version 1.0) (2022). Indian Pharmacopoeia Commission. Indian Pharmacopoeia. Available at: https://ipc.gov.in/images/Bilastine_Tablets_version_1.0.pdf
- Patel, Dr. S., Pasha, Dr. T. Y. (2022). Stability Indicating Isocratic HPLC Method for Bilastine and Characterization of Forced Degradation Products by LC-MS/MS. International Journal of Life Science and Pharma Research, P83–P93. https://doi.org/10.22376/ijlpr.2023.13.1.sp1.p83-p93
- Pathak, B. J., Bhattacharjee, A., Zaman, H., Saikia, H. B., Kapil, M. J., Kumar, S. et al. (2025). New RP-HPLC method for Bilastine estimation in pharmaceutical and bulk dose form. Journal of Chemical Health Risks, 15 (2), 99–105. https://doi.org/10.52783/jchr.v15.i2.7707
- Prathyusha, P., Sundararajan, R., Bhanu, P., Mukthinuthalapati, M. A. (2020). A new stability indicating RP-HPLC method for determination of Bilastine in bulk and pharmaceutical formulation. Research Journal of Pharmacy and Technology, 13 (6), 2849. https://doi.org/10.5958/0974-360x.2020.00507.7
- Chowdary, V. A., Anusha, K, Muneer, S. (2017). Method development and validation of new rp-hplc method for the estimation of bilastine in pharmaceutical dosage form. World Journal of Pharmacy and Pharmaceutical Sciences, 6 (8), 2297–2315. https://doi.org/10.20959/wjpps20178-9923
- Patel, K. K., Patel, A. M., Patel, C. N. (2021). A new simple RP-HPLC Method development, Validation and Forced degradation studies of Bilastine. Asian Journal of Pharmaceutical Analysis, 11 (3), 183–187. https://doi.org/10.52711/2231-5675.2021.00031
- Beltagi, A. M., Lashin, I. A., Essa, W. A., Hathoot, A. A., Azzem, M. A. (2021). Evolution and effectiveness of HPLC Technique for rapid estimation of an Antiallergenic agent Bilastine. Asian Journal of Pharmaceutical Analysis, 11 (2), 57–62. https://doi.org/10.52711/2231-5675.2021.00011
- Bilastine Product Information (2022). Cayman Chemical. Available at: https://cdn.caymanchem.com/cdn/insert/28375.pdf
- Kumar, J., Babra, S., Saini, D., Tiwari, A. (2022). BILASTINE: A Drug Review. International Journal of Innovative Research in Technology, 9 (7), 471–475. Available at: https://ijirt.org/publishedpaper/IJIRT157524_PAPER.pdf
- D Structure. Bromophenol Blue (Compound). PubChem. Available at: https://pubchem.ncbi.nlm.nih.gov/compound/8272#section=Structures
- D Structure. Thymol Blue (Compound). PubChem. Available at: https://pubchem.ncbi.nlm.nih.gov/compound/65565
- Green Solvent Selection Tool. Available at: https://green-solvent-tool.herokuapp.com/
- Derzhavna Farmakopeia Ukrainy. Vol. 1 (2015). Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv». Available at: https://sphu.org/napryamky-diyalnosti/viddil-dfu/dfu-2-0/1-j-tom-dfu-2-0
- Pena-Pereira, F., Wojnowski, W., Tobiszewski, M. (2020). AGREE – Analytical GREEnness Metric Approach and Software. Analytical Chemistry, 92 (14), 10076–10082. https://doi.org/10.1021/acs.analchem.0c01887
- Mansour, F. R., Omer, K. M., Płotka-Wasylka, J. (2024). A total scoring system and software for complex modified GAPI (ComplexMoGAPI) application in the assessment of method greenness. Green Analytical Chemistry, 10, 100126. https://doi.org/10.1016/j.greeac.2024.100126
- Horyn, M., Kryskiw, L., Kucher, T., Zarivna, N., Poliak, O., Logoyda, L. (2025). Novel ecofriendly spectrophotometric methods for the determination of six dihydropyridines calcium channel blockers through derivatization with sulfophtalein dye: application to tablet analysis. BMC Chemistry, 19 (1). https://doi.org/10.1186/s13065-024-01378-x
- Halka, L., Kucher, T., Piponski, M., Kryskiw, L., Zarivna, N., Horyn, M. et al. (2024). Four ecofriendly spectrophotometric methods for the determination of perindopril through derivatization with sulphophtalein dyes: application to tablet analysis. BMC Chemistry, 18 (1). https://doi.org/10.1186/s13065-024-01326-9
- Halka, L., Kucher, T., Kryskiw, L., Piponski, M., Horyn, M., Poliak, O. et al. (2024). Full green assay of rosuvastatin utilizing sulphophtalein dyes: application to tablet analysis. ScienceRise: Pharmaceutical Science, 4 (50), 4–13. https://doi.org/10.15587/2519-4852.2024.310564
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Iryna Ivanusa, Alina-Mariia Horoshko, Anna Staranchuk, Mariya Mykhalkiv

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






