Study of the effect of the microstructure of combined aggregates of nonionic surfactant and cetostearyl alcohol on the rheological properties of hydrophilic cream bases and the release of active substances in experiments in vitro
DOI:
https://doi.org/10.15587/2519-4852.2026.358977Keywords:
cream base, apparent viscosity, aggregate, adsorption layer, surfactant, cetostearyl alcohol, spin probe, EPR spectrum, releaseAbstract
The aim. To study the effect of the microstructure of mixed aggregates and adsorption layers of non-ionic surfactant and cetostearyl alcohol (CSA) on the rheological characteristics of hydrophilic cream bases and the release of certain active substances in experiments in vitro.
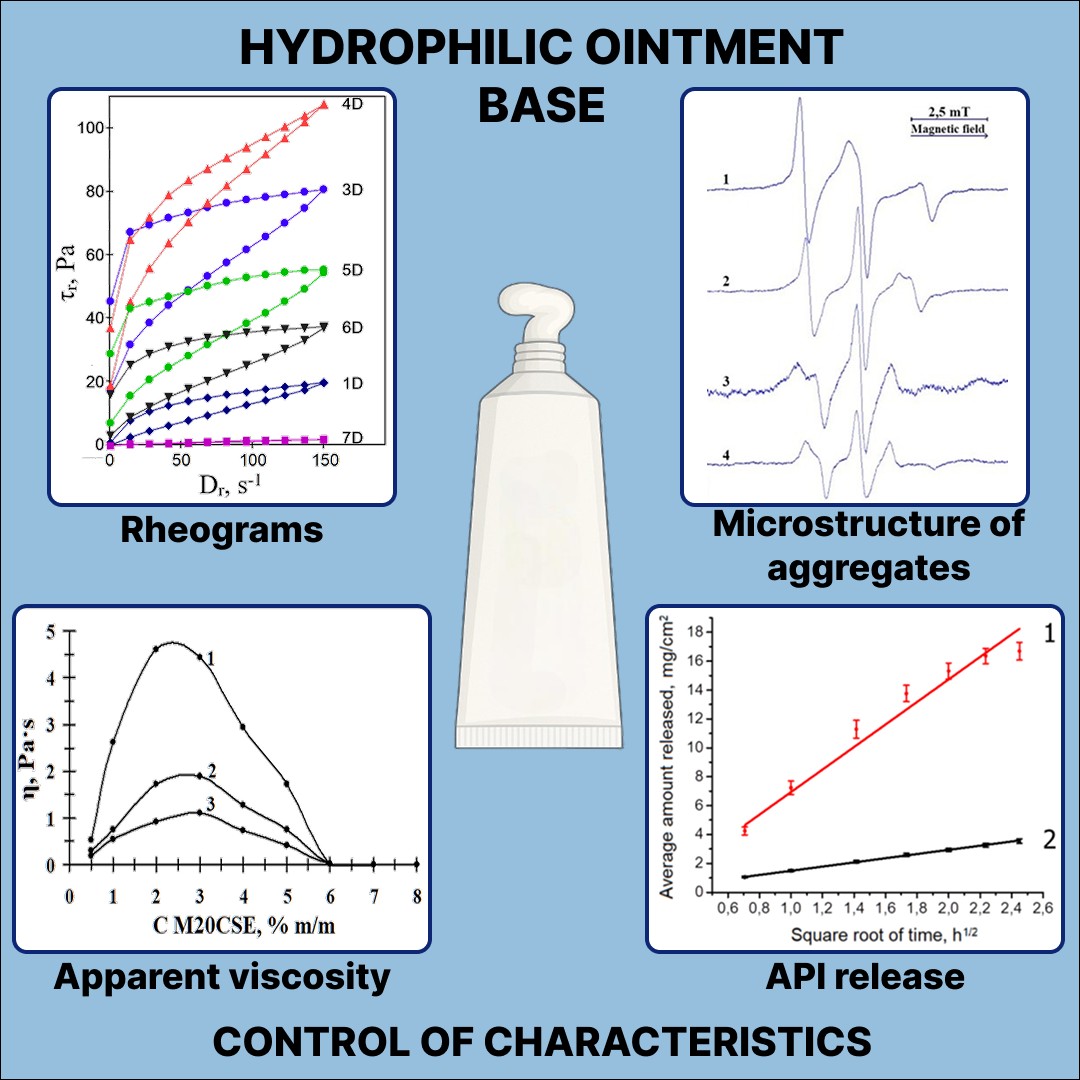
Materials and methods. Cream bases with a dispersion medium of water and propylene glycol (9:1) were studied. The ratio of surfactant to CSA was varied within the base formulations. The rheological properties of the bases were analysed using rotational viscometry, and the microstructure of the aggregates was examined by the spin probe method employing four probes based on fatty acids. The release of active substances from the bases and solutions was tested in vitro; the content of active substances in the dialysate was determined by liquid chromatography.
Results. The apparent viscosity of the bases was observed to reach its maximum at certain ratios of surfactant to CSA, when coagulation structures were formed. The structural arrangement of mixed aggregates of surfactant and CSA was dependent on their ratio. It was demonstrated that a higher specific fraction of CSA in aggregates/adsorption layers contributed to lateral phase separation at the interface with the dispersion medium. This process led to the formation of solid CSA domains and liquid surfactant domains. The mixed aggregates of surfactant and CSA possessed a non-spherical configuration. The hydration of aggregates was ensured by non-ionic surfactant domains. These factors contributed to the formation of coagulation structures at certain concentrations of surfactant and CSA. An increase in the specific fraction of surfactant led to a tendency towards a homogeneous distribution of surfactant and CSA in their aggregates. This was accompanied by a decrease in the apparent viscosity of dispersed systems and a transition from creams to liquids. A decrease in the surfactant fraction resulted in a decline in the hydration of aggregates/adsorption layers, consequently leading to a decrease in the apparent viscosity of the bases. The release of ofloxacin or dexpanthenol was significantly retarded from the bases where coagulation structures were formed.
Conclusions. The rheological characteristics of hydrophilic cream bases are contingent on the microstructure of mixed aggregates or adsorption layers formed by nonionic surfactant and CSA. These properties can be modified by adjusting the mass ratios between these emulsifiers. In the case of cream bases, where a coagulation structure has formed, the release of active ingredients is found to be significantly retarded.
Supporting Agency
- National Academy of Sciences of Ukraine within the framework of the project «Study of dispersed systems with liquid dispersion medium as the primary matrices for medicinal products» (0125U000740)
References
- The European Pharmacopoeia (2022). European Directorate for the Quality of Medicines & HealthCare of the Council of Europe. Strasbourg: Council of Europe, 6106. Available at: http://pheur.edqm.eu/subhome/11-8
- Derzhavna Farmakopeia Ukrainy. Vol. 2 (2024). Kharkiv: Derzhavne pidpryiemstvo «Ukrainskyi naukovyi farmakopeinyi tsentr yakosti likarskykh zasobiv», 424.
- Costa, C., Medronho, B., Filipe, A., Mira, I., Lindman, B., Edlund, H., Norgren, M. (2019). Emulsion Formation and Stabilization by Biomolecules: The Leading Role of Cellulose. Polymers, 11 (10), 1570. https://doi.org/10.3390/polym11101570
- Langevin, D. (2023). Recent Advances on Emulsion and Foam Stability. Langmuir, 39 (11), 3821–3828. https://doi.org/10.1021/acs.langmuir.2c03423
- Lyapunova, A. M., Bezugla, O. P., Lyapunov, O. M. (2017). The study of o/w emulsions using the rotating viscometer method and the method of spin probes. News of Pharmacy, 4 (92), 29–34. https://doi.org/10.24959/nphj.17.2190
- Dekker, R. I., Velandia, S. F., Kibbelaar, H. V. M., Morcy, A., Sadtler, V., Roques-Carmes, T. et al. (2023). Is there a difference between surfactant-stabilised and Pickering emulsions? Soft Matter, 19 (10), 1941–1951. https://doi.org/10.1039/d2sm01375d
- Karishma, S., Rajvanshi, K., Kumar, H., Basavaraj, M. G., Mani, E. (2023). Oil-in-Water Emulsions Stabilized by Hydrophilic Homopolymers. Langmuir, 39 (38), 13430–13440. https://doi.org/10.1021/acs.langmuir.3c00798
- Ataeian, P., Aroyan, L., Parwez, W., Tam, K. C. (2022). Emulsions undergoing phase transition: Effect of emulsifier type and concentration. Journal of Colloid and Interface Science, 617, 214–223. https://doi.org/10.1016/j.jcis.2022.02.140
- Zheng, R., Tian, J., Binks, B. P., Cui, Z., Xia, W., Jiang, J. (2022). Oil-in-Water emulsions stabilized by alumina nanoparticles with organic electrolytes: Fate of particles. Journal of Colloid and Interface Science, 627, 749–760. https://doi.org/10.1016/j.jcis.2022.07.085
- Badruddoza, A. Z. M., Yeoh, T., Shah, J. C., Walsh, T. (2023). Assessing and Predicting Physical Stability of Emulsion-Based Topical Semisolid Products: A Review. Journal of Pharmaceutical Sciences, 112 (7), 1772–1793. https://doi.org/10.1016/j.xphs.2023.03.014
- Binks, B. P. (Ed.) (1998). Modern aspects of Emulsion Science. Royal Society of Chemistry, 442. https://doi.org/10.1039/9781847551474
- Myers, D. (2006). Surfactant Science and Technology. John Wiley & Sons, Inc. https://doi.org/10.1002/047174607X
- Farn, R. J. (Ed.) (2006). Chemistry and Technology of Surfactants. Blackwell Publishing Ltd, 315. https://doi.org/10.1002/9780470988596
- Cai, Z., Wei, Y., Shi, A., Zhong, J., Rao, P., Wang, Q., Zhang, H. (2023). Correlation between interfacial layer properties and physical stability of food emulsions: current trends, challenges, strategies, and further perspectives. Advances in Colloid and Interface Science, 313, 102863. https://doi.org/10.1016/j.cis.2023.102863
- Li, P., Huang, H., Fang, Y., Wang, Y., No, D. S., Bhatnagar, R. S., Abbaspourrad, A. (2023). Interfacial engineering of clear emulsions: Surfactant hydrophobicity and the hidden role of chain structure. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 676, 132242. https://doi.org/10.1016/j.colsurfa.2023.132242
- Botti, T. C., Hutin, A., Quintella, E., Carvalho, M. S. (2022). Effect of interfacial rheology on drop coalescence in water–oil emulsion. Soft Matter, 18(7), 1423–1434. https://doi.org/10.1039/d1sm01382c
- Leister, N., Götz, V., Jan Bachmann, S., Nachtigall, S., Hosseinpour, S., Peukert, W., Karbstein, H. (2023). A comprehensive methodology to study double emulsion stability. Journal of Colloid and Interface Science, 630, 534–548. https://doi.org/10.1016/j.jcis.2022.10.119
- Han, D., Mao, J., Zhao, J., Zhang, H., Wang, D., Cao, H. et al. (2022). Dissipative particle dynamics simulation and experimental analysis of effects of Gemini surfactants with different spacer lengths on stability of emulsion systems. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 655, 130205. https://doi.org/10.1016/j.colsurfa.2022.130205
- McClements, D. J., Jafari, S. M. (2018). Improving emulsion formation, stability and performance using mixed emulsifiers: A review. Advances in Colloid and Interface Science, 251, 55–79. https://doi.org/10.1016/j.cis.2017.12.001
- Hong, I. K., Kim, S. I., Lee, S. B. (2018). Effects of HLB value on oil-in-water emulsions: Droplet size, rheological behavior, zeta-potential, and creaming index. Journal of Industrial and Engineering Chemistry, 67, 123–131. https://doi.org/10.1016/j.jiec.2018.06.022
- Alam, S., Algahtani, M. S., Ahmad, M. Z., Ahmad, J. (2020). Investigation Utilizing the HLB Concept for the Development of Moisturizing Cream and Lotion: In-Vitro Characterization and Stability Evaluation. Cosmetics, 7 (2), 43. https://doi.org/10.3390/cosmetics7020043
- Wang, Q., Zhang, H., Han, Y., Cui, Y., Han, X. (2023). Study on the relationships between the oil HLB value and emulsion stabilization. RSC Advances, 13 (35), 24692–24698. https://doi.org/10.1039/d3ra04592g
- Colafemmina, G., Palazzo, G., Mateos, H., Amin, S., Fameau, A.-L., Olsson, U., Gentile, L. (2020). The cooling process effect on the bilayer phase state of the CTAC/cetearyl alcohol/water surfactant gel. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 597, 124821. https://doi.org/10.1016/j.colsurfa.2020.124821
- Tran, H. H., Nguyen, T. H., Tran, T. T., Vu, H. D., Nguyen, H. M. T. (2021). Structures, Electronic Properties, and Interactions of Cetyl Alcohol with Cetomacrogol and Water: Insights from Quantum Chemical Calculations and Experimental Investigations. ACS Omega, 6 (32), 20975–20983. https://doi.org/10.1021/acsomega.1c02439
- Berliner, L. J., Reuben, J. (Ed.) (1989). Spin Labeling: Theory and Applications. New York: Plenum Press, 670. https://doi.org/10.1007/978-1-4613-0743-3
- Bezuglaya, E., Lyapunov, N., Chebanov, V., Liapunov, O. (2022). Study of the formation of micelles and their structure by the spin probe method. ScienceRise: Pharmaceutical Science, 4 (38), 4–18. https://doi.org/10.15587/2519-4852.2022.263054
- Liapunov, M. O., Ivanov, L. V., Bezugla, O. P., Zhdanov, R. I., Tsymbal, L. V. (1992). Doslidzhennia ahrehativ poverkhnevo-aktyvnykh rechovyn (PAR) metodom spinovykh zondiv. Farmatsevtychnyi zhurnal, 5-6, 40–45.
- Buckingham, R. (Ed.) (2020). Martindale: The Complete Drug Reference, 40th Ed. London: Pharmaceutical Press, 4852.
- Derzhavnyi reiestr likarskykh zasobiv Ukrainy. Available at: http://www.drlz.kiev.ua/
- Sheskey, P. J., Hancock, B. C., Moss, G. P., Goldfarb, D. J. (Eds.) (2020). Handbook of Pharmaceutical Excipients. London: Pharm. Press, 1296.
- Liapunova, A. M., Krasnopyorova, А. P., Bezuglа, О. P., Liapunov, O. M., Yukhnо, G. D., Pukhova, T. М. (2024). Polythermal studies of the water – propylene glycol systems by densitometry, viscometry and spin probes method. Functional Materials, 31 (4), 609–618. https://doi.org/10.15407/fm31.04.609
- Ilić, T., Pantelić, I., Savić, S. (2021). The Implications of Regulatory Framework for Topical Semisolid Drug Products: From Critical Quality and Performance Attributes towards Establishing Bioequivalence. Pharmaceutics, 13 (5), 710. https://doi.org/10.3390/pharmaceutics13050710
- Tiffner, K. I., Kanfer, I., Augustin, T., Raml, R., Raney, S. G., Sinner, F. (2018). A comprehensive approach to qualify and validate the essential parameters of an in vitro release test (IVRT) method for acyclovir cream, 5%. International Journal of Pharmaceutics, 535 (1-2), 217–227. https://doi.org/10.1016/j.ijpharm.2017.09.049
- Lyapunov, N., Bezugla, O., Liapunova, A., Zinchenko, I., Liapunov, O., Lysokobylka, O., Dzhoraieva, S. (2025). Study of some properties of hydrophilic ointment bases depending on their composition. ScienceRise: Pharmaceutical Science, 5 (57), 4–19. https://doi.org/10.15587/2519-4852.2025.339597
- Bezuglaya, E., Liapunova, A., Zinchenko, I., Lyapunov, N. (2023). Study of factors affecting the in vitro release of dexpanthenol from solutions and topical semi-solid preparations. ScienceRise: Pharmaceutical Science, 3 (43), 4–15. https://doi.org/10.15587/2519-4852.2023.279283
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Nikolay Lyapunov, Оlena Bezugla, Oleksii Liapunov, Anna Liapunova, Igor Zinchenko, Yurij Stolper

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






