Pharmacological action of thick extract of common tansy (Tanacetum vulgare L.) flowers in an experimental model of estrogen-induced cholestasis
DOI:
https://doi.org/10.15587/2519-4852.2026.359582Keywords:
estrogen-induced cholestasis (EIC), rats, thick extract of Tanacetum vulgare L. flowers (TETVF), antioxidant properties, protective effectAbstract
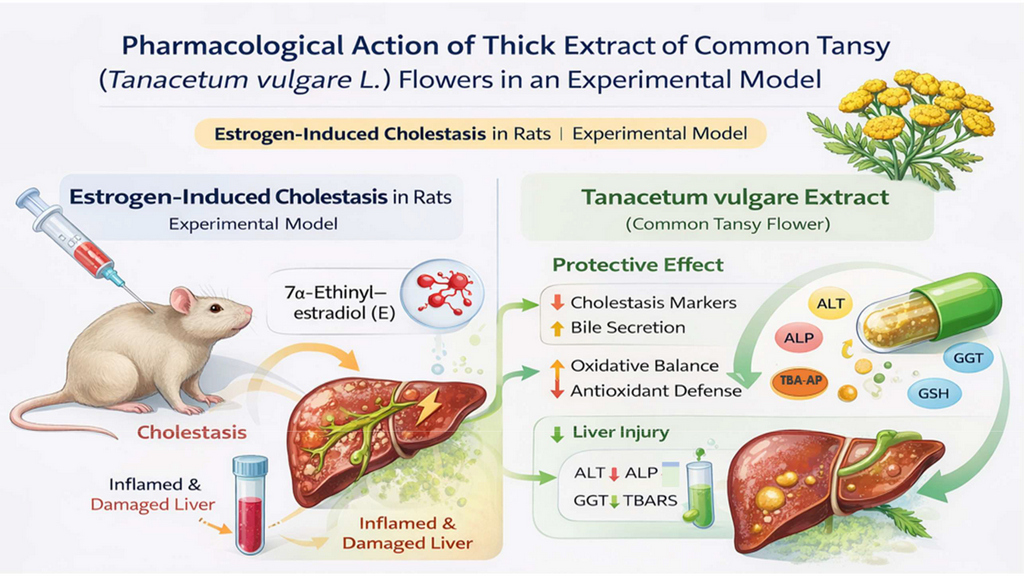
The aim. To study the protective effect of thick extract of Tanacetum vulgare L. flowers (TETVF) in an experimental model of estrogen-induced cholestasis (EIC).
Materials and methods. EIC was reproduced by subcutaneous administration of 7α-ethinylestradiol (E) (5 mg/kg) to rats. TETVF and the reference preparation (RP) cholelesan (ChL) were administered to animals intragastrically once a day for 3 days before and 5 days of the modeling period. One day after the last E administration, the animals were anesthetized (thiopental sodium, 50 mg/kg, intraperitoneally), the volume of bile secreted in 60 min was determined, and the bile secretion rate (BSR) was calculated. The activity of alanine aminotransferase (ALT), alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GTT), the content of TBA-active products (TBA-AP) and reduced glutathione (RG) was determined in the blood serum. For histological examination, prepared liver sections were stained with hematoxylin, eosin and sudan IV to identify lipids. A semi-quantitative assessment of EIC signs was performed.
Results of the study. In animals with EIC TETVF contributed to the normalization of bile volume and BSR to the level of intact control (IC), a significant (p < 0.05) decrease in the studied parameters: ALT, LF and GTT, a decrease in lipid peroxidation (LPO) processes and an increase in liver antioxidant defense, as evidenced by a significant decrease in the level of TBA-AP (30.98±0.90 μmol/g vs. 44.87±4.26, p < 0.05) and a 1.9-fold increase in RG (4.11±0.36 μmol/g vs. 2.19±0.16, p < 0.05) compared to the control pathology (CP). In general, TETVF showed a normalizing effect like RP, but was inferior to it in terms of its effect on BER and on ALT activity and TBA-AP level. After the introduction of TETVF the severity of ductal proliferation significantly decreased by 1.6 times (p < 0.05), periductal inflammation by 3.8 times (p < 0.05), and fatty degeneration of hepatocytes of the periductal zones by 2.3 times (p < 0.05) compared to CP.
Conclusion. The ability of TETVF to improve impaired liver function under conditions of EEIS and reduce pathological manifestations was established
References
- Yu, L., Liu, Y., Wang, S., Zhang, Q., Zhao, J., Zhang, H. et al. (2023). Cholestasis: exploring the triangular relationship of gut microbiota-bile acid-cholestasis and the potential probiotic strategies. Gut Microbes, 15 (1). https://doi.org/10.1080/19490976.2023.2181930
- Kolarić, T. O., Ninčević, V., Smolić, R., Smolić, M., Wu, G. Y. (2019). Mechanisms of Hepatic Cholestatic Drug Injury. Journal of Clinical and Translational Hepatology, 7 (1), 86–92. https://doi.org/10.14218/jcth.2018.00042
- Onofrio, F. Q., Hirschfield, G. M. (2020). The Pathophysiology of Cholestasis and Its Relevance to Clinical Practice. Clinical Liver Disease, 15 (3), 110–114. https://doi.org/10.1002/cld.894
- Yu, L., Liu, X., Li, X., Yuan, Z., Yang, H., Zhang, L., Jiang, Z. (2016). Protective effects of SRT1720 via the HNF1α/FXR signalling pathway and anti-inflammatory mechanisms in mice with estrogen-induced cholestatic liver injury. Toxicology Letters, 264, 1–11. https://doi.org/10.1016/j.toxlet.2016.10.016
- Chen, C., Gong, X., Yang, X., Shang, X., Du, Q., Liao, Q. et al. (2019). The roles of estrogen and estrogen receptors in gastrointestinal disease (Review). Oncology Letters, 18 (6), 5673–5680. https://doi.org/10.3892/ol.2019.10983
- Smith, D. D., Rood, K. M. (2020). Intrahepatic Cholestasis of Pregnancy. Clinical Obstetrics & Gynecology, 63 (1), 134–151. https://doi.org/10.1097/grf.0000000000000495
- Mutlu, M. F., Aslan, K., Guler, I., Mutlu, I., Erdem, M., Bozkurt, N., Erdem, A. (2017). Two cases of first onset intrahepatic cholestasis of pregnancy associated with moderate ovarian hyperstimulation syndrome after IVF treatment and review of the literature. Journal of Obstetrics and Gynaecology, 37 (5), 547–549. https://doi.org/10.1080/01443615.2017.1286302
- Mor, M., Shmueli, A., Krispin, E., Bardin, R., Sneh-Arbib, O., Braun, M. et al. (2020). Intrahepatic cholestasis of pregnancy as a risk factor for preeclampsia. Archives of Gynecology and Obstetrics, 301 (3), 655–664. https://doi.org/10.1007/s00404-020-05456-y
- Ma, X., Jiang, Y., Zhang, W., Wang, J., Wang, R., Wang, L. et al. (2020). Natural products for the prevention and treatment of cholestasis: A review. Phytotherapy Research, 34 (6), 1291–1309. https://doi.org/10.1002/ptr.6621
- Wang, R., Cheng, N., Peng, R., Yu, Z., Nan, M., Cao, H. (2020). Oral herbal medicine for women with intrahepatic cholestasis in pregnancy: a systematic review of randomized controlled trials. BMC Complementary Medicine and Therapies, 20 (1). https://doi.org/10.1186/s12906-020-03097-x
- Mishchenko, O. Ya., Hontova, T. M., Yurchenko, K. Yu., Zolotaikina, M. Yu., Herbina, N. A., Ruban, O. A., Kalko, K. O. (2020). Pat. 139684 UA. Analhetychnyi Zasib. MKP (2006) A61K 36/00, A61K 9/02 (2006.01) A61R 29/00. No. u201907609; declareted: 08.07.2019; published:10.01.2020, Bul. No. 1/2020.
- Herbina, N. A., Ruban, O. A., Gontova, T. M., Yaremenko, M. S., Yurchenko, C. Yu. (2021). The study of the qualitative and quantitative content of the amount of flavonoids and hydroxycinnamic acids in a dense extract of common tansy flowers. News of Pharmacy, 2 (102), 8–13. https://doi.org/10.24959/nphj.21.55
- Yurchenko, C., Mishchenko, O., Herbina, N., Каpustyanskiy, I. (2021). Screening study of the choleretic and cholekinetic effects of the the common tansy (Tanacetum vulgare L.) flowers thick extract. Pharmacology and Drug Toxicology, 15 (3), 197–203. https://doi.org/10.33250/15.03.197
- Xu, Y.-J., Yu, Z.-Q., Zhang, C.-L., Li, X.-P., Feng, C.-Y., Lei, K. et al. (2025). CORRIGENDUM: Protective Effects of Ginsenosides on 17α-Ethynyelstradiol-Induced Intrahepatic Cholestasis via Anti-Oxidative and Anti-Inflammatory Mechanisms in Rats. The American Journal of Chinese Medicine, 53 (8), 2603–2604. https://doi.org/10.1142/s0192415x2592003x
- Kholelesan® kapsuly blister No. 30. Compendium. Available at: https://compendium.com.ua/dec/338632/446070/
- Drohovoz, S. M., Hubskyi, Yu. I., Skakun, M. P., Kovalenko, V. M., Derymedvid, L. V. (2001). Eksperymentalne vyvchennia zhovchohinnoi, kholespazmolitychnoi, kholelitiaznoi ta hepatoprotektornoi aktyvnosti novykh likarskykh zasobiv. Doklinichne doslidzhennia likarskykh zasobiv. Kyiv, 334–351.
- European convention for the protection of vertebrate animals used for the experimental and other scientific purposes: European Treaty Series No. 123: Text amended according to the provisions of the Protocol (ETS No. 170), as of its entry into force, on 2 December 2005 (1986). Strasbourg, 48.
- Beutler, E., Duron, O., Kelly, B. M. (1963). Improved method for the determination of blood glutathione. Journal of Laboratory and Clinical Medicine, 61, 882–888.
- Golovanova, I. A., Belikova, I. V., Lyakhova, N. O. (2017). Osnovy medychnoi statystyky. Poltava, 113.
- Crocenzi, F. A., Sánchez Pozzi, E. J., Pellegrino, J. M., Favre, C. O., Rodríguez Garay, E. A., Mottino, A. D. et al. (2001). Beneficial Effects of Silymarin on Estrogen–Induced Cholestasis in the Rat: A Study In Vivo And in Isolated Hepatocyte Couplets. Hepatology, 34 (2), 329–339. https://doi.org/10.1053/jhep.2001.26520
- Zu, Y., Yang, J., Zhang, C., Liu, D. (2021). The Pathological Mechanisms of Estrogen-Induced Cholestasis: Current Perspectives. Frontiers in Pharmacology, 12. https://doi.org/10.3389/fphar.2021.761255
- Kawamura, K., Kobayashi, Y., Kageyama, F., Kawasaki, T., Nagasawa, M., Toyokuni, S. et al. (2000). Enhanced Hepatic Lipid Peroxidation in Patients With Primary Biliary Cirrhosis. American Journal of Gastroenterology, 95 (12), 3596–3601. https://doi.org/10.1111/j.1572-0241.2000.03300.x
- Hussein, M. A. (2013). Prophylactic Effect of Resveratrol Against Ethinylestradiol-Induced Liver Cholestasis. Journal of Medicinal Food, 16 (3), 246–254. https://doi.org/10.1089/jmf.2012.0183
- Mureşana, M., Benedeca, D., Vlasea, L., Opreana, R., Toiua, A., Onigaa, I. (2015). Screening of polyphenolic compounds, antioxidant and antimicrobial properties of Tanacetum vulgare from Transylvania. Studia Universitatis Babes-Bolyai Chemia, 60 (1), 127–138.
- Mishchenko, O., Kyrychenko, I., Gontova, T., Kalko, K., Hordiei, K. (2022). Research on the phenolic profile, antiradical and anti-inflammatory activity of a thick hydroalcoholic feverfew (Tanacetum parthenium L.) herb extract. ScienceRise: Pharmaceutical Science, 5 (39), 91–99. https://doi.org/10.15587/2519-4852.2022.266400
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Oksana Mishchenko, Yaroslava Butko, Oksana Tkachova, Olena Khalieieva, Andrii Berezniakov, Oleksii Andriianenkov

This work is licensed under a Creative Commons Attribution 4.0 International License.
Our journal abides by the Creative Commons Attribution 4.0 International License copyright rights and permissions for open access journals.
Authors, who are published in this journal, agree to the following conditions:
1. The authors reserve the right to authorship of the work and pass the first publication right of this work to the journal under the terms of a Creative Commons Attribution 4.0 International License, which allows others to freely distribute the published research with the obligatory reference to the authors of the original work and the first publication of the work in this journal.
2. The authors have the right to conclude separate supplement agreements that relate to non-exclusive work distribution in the form in which it has been published by the journal (for example, to upload the work to the online storage of the journal or publish it as part of a monograph), provided that the reference to the first publication of the work in this journal is included.
3. Authors have the right to store the final accepted version of the article in an institutional, thematic, or any other repository to ensure visibility and accessibility.






