Структурна модифікація 1,2,4-тріазольного ядра як стратегія створення біологічно активних сполук (Огляд літератури)
DOI:
https://doi.org/10.15587/2519-4852.2026.356807Ключові слова:
1,2,4-тріазол, хімічна модифікація, біологічна активність, синтез, фармакофор, алкілування, гібридні сполуки, молекулярний докінг, ADME-аналіз, SARАнотація
Мета. Метою оглядової статті є систематизація та узагальнення сучасних літературних даних щодо методів хімічної модифікації похідних 1,2,4-тріазолу, а також аналіз впливу структурних перетворень на їх біологічну активність і фармакологічний потенціал.
Матеріали і методи. Об’єктом аналізу слугували наукові публікації вітчизняних та зарубіжних авторів, присвячені синтезу, функціоналізації та біологічному дослідженню похідних 1,2,4-тріазолу. Узагальнення даних здійснювали з використанням методів системного аналізу, порівняльної оцінки синтетичних підходів, а також аналізу результатів in silico, in vitro та in vivo досліджень (молекулярний докінг, ADME-аналіз, SAR-оцінка).
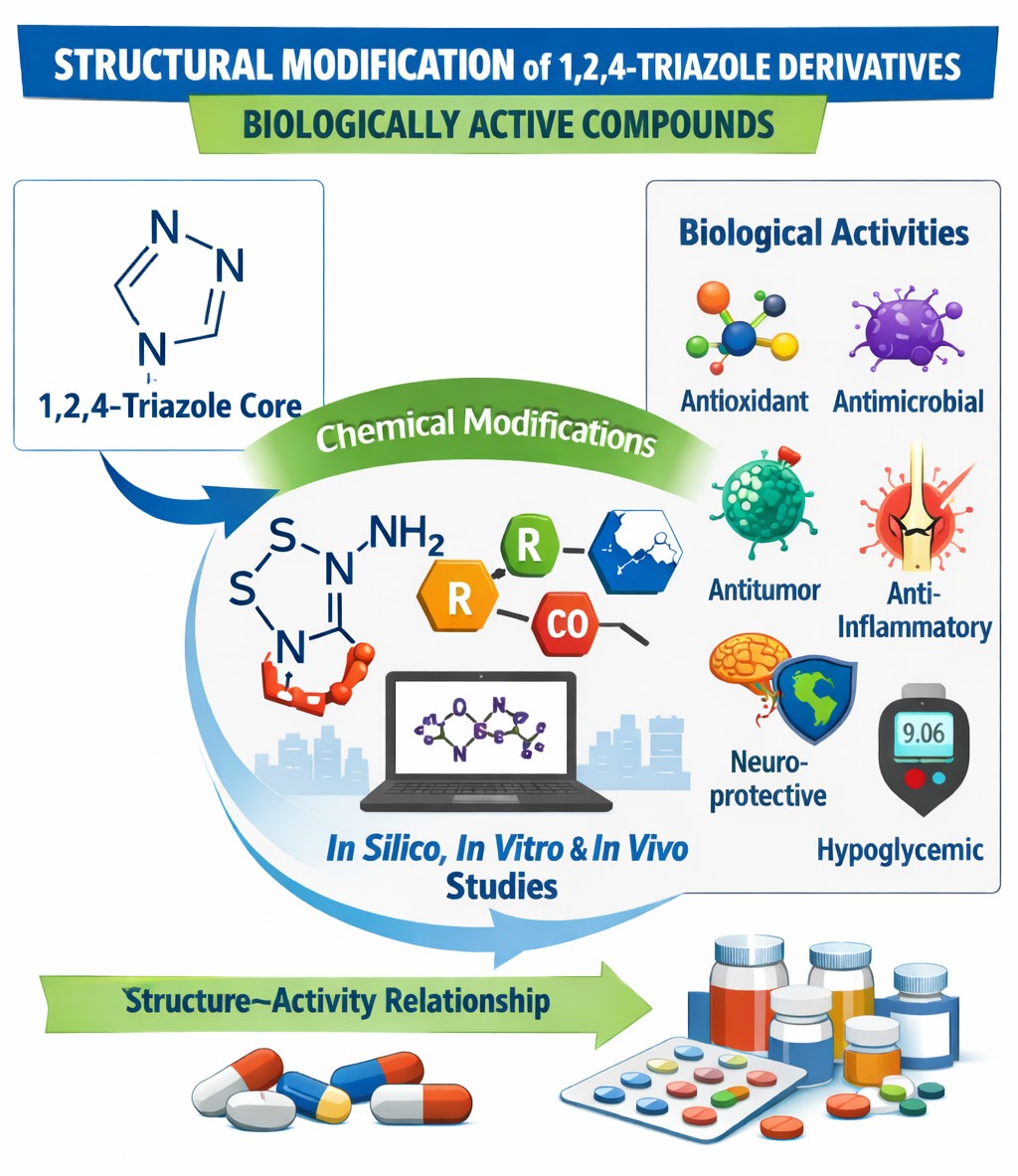
Результати. Показано, що похідні 1,2,4-тріазолу характеризуються високою хімічною лабільністю та здатністю до модифікації за атомом сірки, аміногрупою і атомами азоту гетероциклічного ядра. Реакції алкілування, ацилювання, утворення солей, гібридизації з іншими фармакофорними фрагментами, а також застосування мікрохвильового синтезу дозволяють отримувати сполуки з широким спектром біологічної дії. Серед досліджених похідних виявлено речовини з антиоксидантною, антимікробною, протипухлинною, протизапальною, нейропротекторною та цукрознижуючою активністю. Встановлено кореляцію між хімічною будовою сполук, характером замісників та їх фармакологічними властивостями.
Висновки. Похідні 1,2,4-тріазолу є перспективною фармакофорною платформою для створення нових біологічно активних сполук. Подальше цілеспрямоване вивчення напрямів їх хімічної модифікації та залежності структура-активність відкриває широкі можливості для розробки потенційних лікарських засобів
Спонсор дослідження
- Research project "Creation of 1,2,4-triazole derivatives with polypharmacological activity for the needs of military medicine and combat victims" with registration number 0126U001480 and This study was funded by European Union-NextGenerationEU, through the National Recovery and Resilience Plan of the Republic of Bulgaria, project № BG-RRP-2.004-0009-C02
Посилання
- Hashem, H. E., Amr, A. E.-G. E., Nossier, E. S., Anwar, M. M., Azmy, E. M. (2022). New Benzimidazole-, 1,2,4-Triazole-, and 1,3,5-Triazine-Based Derivatives as Potential EGFRWT and EGFRT790M Inhibitors: Microwave-Assisted Synthesis, Anticancer Evaluation, and Molecular Docking Study. ACS Omega, 7 (8), 7155–7171. https://doi.org/10.1021/acsomega.1c06836
- Emami, L., Sadeghian, S., Mojaddami, A., Khabnadideh, S., Sakhteman, A., Sadeghpour, H. et al. (2022). Design, synthesis and evaluation of novel 1,2,4-triazole derivatives as promising anticancer agents. BMC Chemistry, 16 (1). https://doi.org/10.1186/s13065-022-00887-x
- Dai, J., Tian, S., Yang, X., Liu, Z. (2022). Synthesis methods of 1,2,3-/1,2,4-triazoles: A review. Frontiers in Chemistry, 10. https://doi.org/10.3389/fchem.2022.891484
- Soldi, L. R., Oliveira, A. P. de L., Silva, M. J. B. (2026). Emerging Applications of Triazole Antifungal Drugs. International Journal of Molecular Sciences, 27 (2), 817. https://doi.org/10.3390/ijms27020817
- Demchenko, N., Suvorova, Z., Fedchenkova, Y., Shpychak, T., Shpychak, O., Bobkova, L., Demchenko, S. (2021). Synthesis and antibacterial activity of 3-arylaminomethyl-1-(2-oxo-2-arylethyl)-6,7,8,9-tetrahydro-5H-[1,2,4]triazolo[4,3-a] azepin-1-ium bromides and aryl-(4-R1-phenyl-5,6,7,8-tetrahydro-2,2a,8a-triazacyclopenta[cd]azulen-1-ylmethyl)-amines. ScienceRise: Pharmaceutical Science, 6 (34), 51–57. https://doi.org/10.15587/2519-4852.2021.249480
- Beyzaei, H., Ghanbari Kudeyani, M., Samareh Delarami, H., Aryan, R. (2020). Synthesis, antimicrobial and antioxidant evaluation, and molecular docking study of 4,5-disubstituted 1,2,4-triazole-3-thiones. Journal of Molecular Structure, 1215, 128273. https://doi.org/10.1016/j.molstruc.2020.128273
- Mahadev, J. K., Nandkumar, C. A., Vishveswar, K. A., Salve, P. L., Pise, V. J. (2024). 1,3,4- Thiadiazole and Its Derivatives- A Review on Syntheic Account and Recent Progress on its Phermoacological Activities. Asian Journal of Pharmaceutical Research and Development, 12 (3), 129–143. https://doi.org/10.22270/ajprd.v11i3.1406
- Tariq, S., Alam, O., Mohd, A. (2018). Synthesis, anti-inflammatory, p38α MAP kinase inhibitory activities and molecular docking studies of quinoxaline derivatives containing triazole moiety. Bioorganic Chemistry, 76, 343–358. https://doi.org/10.1016/j.bioorg.2017.12.003
- Ladgaonkar, Y. A., Waskar, P. (2025). QSAR Study of 1, 2, 4-Triazole for their Anticancer Activity. Asian Journal of Research in Chemistry, 18 (3), 117–122. https://doi.org/10.52711/0974-4150.2025.00019
- Taieb Brahimi, F., Belkhadem, F., Trari, B., Othman, A. A. (2020). Diazole and triazole derivatives of castor oil extract: synthesis, hypoglycemic effect, antioxidant potential and antimicrobial activity. Grasas y Aceites, 71 (4), e378. https://doi.org/10.3989/gya.0342191
- Yildirim, N. (2020). Synthesis of Novel 3-(4-tert-Butylphenyl)-5-Cylopropyl-4H-1,2,4-Triazole Derivatives with Antioxidative and Antimicrobial Activities. Letters in Drug Design & Discovery, 17 (8), 954–964. https://doi.org/10.2174/1570180817999200424073524
- Khokhar, D., Jadoun, S., Arif, R., Jabin, S., Budhiraja, V. (2021). Copolymerization of o-phenylenediamine and 3-Amino-5-methylthio-1H-1,2,4-triazole for tuned optoelectronic properties and its antioxidant studies. Journal of Molecular Structure, 1228, 129738. https://doi.org/10.1016/j.molstruc.2020.129738
- Geetha, B. M., Brinda, K. N., Achar, G., Małecki, J. G., Alwarsamy, M., Betageri, V. S., Budagumpi, S. (2020). Coumarin incorporated 1,2,4–triazole derived silver(I) N–heterocyclic carbene complexes as efficient antioxidant and antihaemolytic agents. Journal of Molecular Liquids, 301, 112352. https://doi.org/10.1016/j.molliq.2019.112352
- Koval, A., Lozynskyi, A., Shtrygol’, S., Lesyk, R. (2022). An overview on 1,2,4-triazole and 1,3,4-thiadiazole derivatives as potential anesthesic and anti-inflammatory agents. ScienceRise: Pharmaceutical Science, 2 (36), 10–17. https://doi.org/10.15587/2519-4852.2022.255276
- Eya’ane Meva, F., Prior, T. J., Evans, D. J., Shah, S., Tamngwa, C. F., Belengue, H. G. L. et al. (2021). Anti-inflammation and antimalarial profile of 5-pyridin-2-yl-1H-[1,2,4]triazole-3-carboxylic acid ethyl ester as a low molecular intermediate for hybrid drug synthesis. Research on Chemical Intermediates, 48 (2), 885–898. https://doi.org/10.1007/s11164-021-04607-3
- Shcherbyna, R. (2020). An investigation of the pharmacokinetics and potential metabolites of potassium 2-((4-amino-5-(morfolinometyl)-4H-1,2,4-triazol-3-yl)thio)acetate on rats. Journal of Faculty of Pharmacy of Ankara University, 44 (2), 233–241. https://doi.org/10.33483/jfpau.681611
- Sirakanyan, S. N., Spinelli, D., Geronikaki, A., Hakobyan, E. K., Petrou, A., Kartsev, V. G. et al (2024). New triazole-based hybrids as neurotropic agents. RSC Advances, 14 (45), 32922–32943. https://doi.org/10.1039/d4ra06121g
- Frolova, Y., Kaplaushenko, A., Sameliuk, Y., Romanina, D., Morozova, L. (2022). Investigation of the antimicrobial and antifungal activities of some 1,2,4-triazole derivatives. Česká a Slovenská Farmacie, 71 (4), 149–158. https://doi.org/10.5817/csf2022-4-149
- Slivka, M., Fizer, M., Mariychuk, R., Ostafin, M., Moyzesh, O., Koval, G. et al. (2022). Synthesis and Antimicrobial Activity of Functional Derivatives of thiazolo[ 2,3-c][1,2,4]triazoles. Letters in Drug Design & Discovery, 19 (9), 791–799. https://doi.org/10.2174/1570180819666220110145659
- Abulkhair, H. S. (2025). α-Glucosidase-targeting 1,2,4-triazole antidiabetic candidates: comparative analysis and future perspectives. Future Medicinal Chemistry, 17 (24), 3025–3040. https://doi.org/10.1080/17568919.2025.2587561
- Sargsyan, S., Sargsyan, A., Khizantsyan, K., Aghajanyan, I., Sargsyan, T., Margaryan, K., Pompella, A. (2023). Electrosynthesis of silver metallonanocomposites in the 1-vinyl-1.2.4-triazole copolymer matrix with methacrylic acid. Polymers and Polymer Composites, 31. https://doi.org/10.1177/09673911231166640
- Meduri, B., Pavan, S. R., Prabhu, A., Shankaranarayana, A. H., Sethu, A. K., Singh, M. et al. (2024). New LPA1 receptor modulators: Design, synthesis, in-silico, and anticancer studies of triazole and oxadiazole analogs. Journal of Molecular Structure, 1295, 136672. https://doi.org/10.1016/j.molstruc.2023.136672
- Crider, A. M., Hospital, A., Sandoval, K. E., Neumann, W. L., Kukielski, S., Garic, L. et al. (2025). 3-Thio-3,4,5-trisubstituted-1,2,4-triazoles: high affinity somatostatin receptor-4 agonist synthesis and structure–activity relationships. RSC Medicinal Chemistry, 16 (2), 945–960. https://doi.org/10.1039/d4md00597j
- Węglińska, L., Bekier, A., Trotsko, N., Kaproń, B., Plech, T., Dzitko, K., & Paneth, A. (2022). Inhibition of Toxoplasma gondii by 1,2,4-triazole-based compounds: marked improvement in selectivity relative to the standard therapy pyrimethamine and sulfadiazine. Journal of Enzyme Inhibition and Medicinal Chemistry, 37 (1), 2621–2634. https://doi.org/10.1080/14756366.2022.2112576
- Bağlan, M., Gören, K., Yildiko, Ü. (2023). DFT Computations and Molecular Docking Studies of 3-(6-(3-aminophenyl)thiazolo[1,2,4]triazol-2-yl)-2H-chromen-2-one(ATTC) Molecule. Hittite Journal of Science and Engineering, 10 (1), 11–19. https://doi.org/10.17350/hjse19030000286
- Favre, H. A., Powell, W. H. (Eds.) (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred IUPAC Names 2013. Royal Society of Chemistry. Available at: https://iupac.qmul.ac.uk/BlueBook/contents.html
- Leigh, J. (Ed.) (2011). Principles of Chemical Nomenclature: A Guide to IUPAC Recommendations. RSC Publishing. Available at: https://iupac.org/what-we-do/books/principles/
- Kaiser, J., Hibbert, D. B., Stohner, J. (2022). Preparation, formatting and review of IUPAC Technical Reports and Recommendations, IUPAC-sponsored books, or other items carrying the IUPAC label. Pure and Applied Chemistry, 94 (11-12), 1257–1267. https://doi.org/10.1515/pac-2022-1106
- IUPAC Color Books: authoritative resource for chemical nomenclature and terminology. IUPAC.org. Available at: https://iupac.org/what-we-do/books/color-books/
- Trofimova, T. S., Hotsulia, A. S. (2023). Alkyl derivatives of 7’-((3-mercapto-4-methyl-1,2,4-triazol-5-yl)methyl)-3-methylxanthine: synthesis and properties. Farmatsevtychnyi Zhurnal, 5, 45–52. https://doi.org/10.32352/0367-3057.5.23.05
- Dovbnya, D. V., Kaplaushenko, A. G., Korzhova, A. S. (2021). Synthesis and alkylation of 5-aryl-1,2-dihydro-3H-1,2,4-triazole-3-thiones. Journal of Organic and Pharmaceutical Chemistry, 19 (2 (74)), 53–59. https://doi.org/10.24959/ophcj.21.188135
- Bakumovska, Kh. D., Hotsulia, A. S. (2023). Synthesis and properties of 7-((6-aryl-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazol-3-yl)methyl)theophyllines. Farmatsevtychnyi Zhurnal, 1, 58–65. https://doi.org/10.32352/0367-3057.1.23.06
- Hotsulia, A. S., Fedotov, S. O. (2020). Synthesis and properties of 5-(((5-amino-1,3,4-thiadiazole-2-yl)thio)methyl)-4-phenyl-1,2,4-triazole-3-thione and its some S-derivatives. Current Issues in Pharmacy and Medicine: Science and Practice, 33 (2), 182–186. https://doi.org/10.14739/2409-2932.2020.2.207062
- Sameliuk, Y., Isaycheva, K., Kaplaushenko, A. (2024). Synthesis, determination of physico-chemical parameters, structure confirmation, and antioxidant activity of compounds based on 3,5-bis(5-mercapto-4-R-4Н-1,2,4-triazole-3-yl)phenol. ScienceRise: Pharmaceutical Science, 5 (51), 63–70. https://doi.org/10.15587/2519-4852.2024.313832
- Dovbnia, D. V., Kaplaushenko, A. H. (2023). Methods of synthesis of 1,2,4-triazole derivatives with methoxyphenyl and study the spectrum of their pharmacological activity. Current Issues in Pharmacy and Medicine: Science and Practice, 16 (1), 66–73. https://doi.org/10.14739/2409-2932.2023.1.267174
- Oderinlo, O. O., Jordaan, A., Seldon, R., Isaacs, M., Hoppe, H. C., Warner, D. F. et al. (2023). Hydrazone‐Tethered 5‐(Pyridin‐4‐yl)‐4H‐1,2,4‐triazole‐3‐thiol Hybrids: Synthesis, Characterisation, in silico ADME Studies, and in vitro Antimycobacterial Evaluation and Cytotoxicity. ChemMedChem, 18 (6), 143–172. https://doi.org/10.1002/cmdc.202200572
- Dovbnia, D. V., Kaplaushenko, A. H., Frolova, Yu. S. (2021). Synthesis and transformation in the series of 2-((5-(2,4- and 3,4-dimethoxyphenyl)-3H-1,2,4-triazole-3-yl)thio)acetic acids. Current Issues in Pharmacy and Medicine: Science and Practice, 14 (1), 12–16. https://doi.org/10.14739/2409-2932.2021.1.226741
- Pachuta-Stec, A. (2022). Antioxidant Activity of 1,2,4-Triazole and its Derivatives: A Mini-Review. Mini-Reviews in Medicinal Chemistry, 22 (7), 1081–1094. https://doi.org/10.2174/1389557521666210401091802
- Abdelrehim, E. M. (2021). Synthesis and Screening of New [1,3,4]Oxadiazole, [1,2,4]Triazole, and [1,2,4]Triazolo[4,3-b][1,2,4]triazole Derivatives as Potential Antitumor Agents on the Colon Carcinoma Cell Line (HCT-116). ACS Omega, 6 (2), 1687–1696. https://doi.org/10.1021/acsomega.0c05718
- Paprocka, R., Kołodziej, P., Wiese-Szadkowska, M., Helmin-Basa, A., Bogucka-Kocka, A. (2022). Evaluation of Anthelmintic and Anti-Inflammatory Activity of 1,2,4-Triazole Derivatives. Molecules, 27 (14), 4488. https://doi.org/10.3390/molecules27144488
- Fedotov, S. O., Hotsulia, A. S. (2021). Synthesis and properties of S-derivatives of 4-amino-5-(5-methylpyrazol-3-yl)-1,2,4-triazole-3-thiol. Current Issues in Pharmacy and Medicine: Science and Practice, 14 (3), 268–274. https://doi.org/10.14739/2409-2932.2021.3.243176
- Dovbnia, D. V., Kaplaushenko, A. H., Salionov, V. O. (2025). Study of modern synthesis methods of 1,2,4-triazole derivatives, their reactivity, and pharmacological efficacy (Literature review). Farmatsevtychnyi Zhurnal, 4, 28–48. https://doi.org/10.32352/0367-3057.4.25.03
- Gotsulya, А. (2020). Synthesis and antiradical activity of alkyl derivatives of 5-(5-methyl-1H-pyrazol-3-yl)-4-phenyl-4H-1,2,4-triazole-3-thiol. Ankara Universitesi Eczacilik Fakultesi Dergisi, 44 (2), 211–219. https://doi.org/10.33483/jfpau.616116
- Gotsulya, А., Fedotov, S., Zinych, O., Trofimova, T., Brytanova, T. (2023). Synthesis and properties of S-alkyl 4-(4-chlorophenyl)-5-(pyrrole-2-yl)-1,2,4-triazole-3-thiol derivatives. Ankara Universitesi Eczacilik Fakultesi Dergisi, 47 (3). https://doi.org/10.33483/jfpau.1280492
- Zvenihorodska, T., Hotsulia, A., Kravchenko, S., Fedotov, S., Kyrychko, B. (2021). Synthesis and Antimicrobial Action of 1,2,4-Triazole Derivatives Containing Theophylline and 1,3,4-Thiadiazole Fragments In their Structure. African Journal of Biomedical Research, 24 (1), 159–163. https://doi.org/10.4314/
- Safonov, A., Nevmyvaka, A., Panasenko, O., Knysh, Y. (2021). Microwave synthesis of 3- and 4-substituted-5-((3-phenylpropyl)thio)-4H-1,2,4-triazoles. Ankara Universitesi Eczacilik Fakultesi Dergisi, 45 (3), 457–466. https://doi.org/10.33483/jfpau.902274
- Safonov, A. (2020). Microwave synthesis of new 3-(alkylthio)-5-(thiophen-2-ylmethyl)-1,2,4-triazol-4-amines. Journal of Faculty of Pharmacy of Ankara University, 44 (1), 89–98. https://doi.org/10.33483/jfpau.620599
- Safonov, A., Demianenko, D., Vashchyk, Ye., Larianovska, Yu., Lytkin, D., Shcherbyna, R. et al. (2022). Histological study of a corrective influence of sodium 2-((4-amino-5-(thiophen-2-ylmethyl)-4H-1,2,4-triazol-3-yl)thio)acetate on the state of rats liver under conditions of acute immobilization stress. Ankara Universitesi Eczacilik Fakultesi Dergisi, 46 (2), 330–341. https://doi.org/10.33483/jfpau.1012893
- Usenko, D., Kaplaushenko, A. (2024). Study of stress degradation and determination of the stability of the substance and injection solution of sodium 2-((4-amino-5-thiophen-2-ylmethyl)-4H-1,2,4-triazol-3-yl)thio)acetate. The Odesa Medical Journal, 2, 85–92. https://doi.org/10.32782/2226-2008-2024-2-15
- Safonov, A., Gotsulya, A., Shcherbyna, R., Panasenko, O. (2025). Analysis of the effect of sodium 2-((4-amino-5-(thiophen-2-ylmethyl)-4H-1,2,4-triazol-3-yl)thio)acetate high doses on brain tissue. The Odessa Medical Journal, 1, 80–83. https://doi.org/10.32782/2226-2008-2025-1-15
- Shcherbyna, R., Panasenko, O., Polonets, O., Nedorezaniuk, N., Duchenko, M. (2021). Synthesis, antimicrobial and antifungal activity of ylidenhydrazides of 2-((4-R-5-R1-4H-1,2,4-triazol-3-yl)thio)acetaldehydes. Ankara Universitesi Eczacilik Fakultesi Dergisi, 45 (3), 504–514. https://doi.org/10.33483/jfpau.939418
- Kalchenko, V., Shcherbyna, R., Panasenko, O., Salionov, V., Morozova, L. (2024). Evaluation of the antioxidant potential of some 5-(2-bromo-4-fluorophenyl)-4-ethyl-1,2,4-triazole-3-thiol derivatives. The Odessa Medical Journal, 5, 89–94. https://doi.org/10.32782/2226-2008-2024-5-15
- Shcherbyna, R., Kalchenko, V., Kulish, S., Salionov, V., Morozova, L., Nedorezaniuk, N., Mazur, O. (2023). Synthesis, characterization, molecular docking studies of new alkyl derivatives of 5-(2-bromo-4-fluorophenyl)-4-ethyl-4H-1,2,4-triazole-3-thiol. Česká a Slovenská Farmacie, 72 (4), 190–200. https://doi.org/10.36290/csf.2023.004
- Shcherbyna, R., Pruhlo, Y., Duchenko, M., Kulagina, M., Kudria, V. (2022). Evaluation of antioxidant activity of 1,2,4-triazole derivatives with morpholine moiety. Hacettepe University Journal of the Faculty of Pharmacy, 42 (2), 44–58. https://doi.org/10.52794/hujpharm.1033112
- Dovbnia, D., Frolova, Y., Kaplaushenko, A. (2023). A study of hypoglycemic activity of acids and salts containing 1,2,4-triazole. Česká a Slovenská Farmacie, 72 (3), 113–124. https://doi.org/10.5817/csf2023-3-113
- Dovbnya, D. V., Kaplaushenko, A. H., Frolova, Y. S., Pruglo, E. S. (2022). Synthesis and antioxidant properties of new (2,4- and 3,4-dimethoxyphenyl)-1,2,4-triazoles. Pharmacia, 69 (1), 135–142. https://doi.org/10.3897/pharmacia.69.e74107
- Mahmoud, E., Abdelhamid, D., Youssif, B. G. M., Gomaa, H. A. M., Hayallah, A. M., Abdel‐Aziz, M. (2024). Design, synthesis, and antiproliferative activity of new indole/1,2,4‐triazole/chalcone hybrids as EGFR and/or c‐MET inhibitors. Archiv Der Pharmazie, 357 (9). https://doi.org/10.1002/ardp.202300562
- Shahzadi, I., Zahoor, A. F., Rasul, A., Mansha, A., Ahmad, S., Raza, Z. (2021). Synthesis, Hemolytic Studies, and In Silico Modeling of Novel Acefylline–1,2,4-Triazole Hybrids as Potential Anti-cancer Agents against MCF-7 and A549. ACS Omega, 6 (18), 11943–11953. https://doi.org/10.1021/acsomega.1c00424
- Akhter, N., Batool, S., Khan, S. G., Rasool, N., Anjum, F., Rasul, A. et al. (2023). Bio-Oriented Synthesis and Molecular Docking Studies of 1,2,4-Triazole Based Derivatives as Potential Anti-Cancer Agents against HepG2 Cell Line. Pharmaceuticals, 16 (2), 211. https://doi.org/10.3390/ph16020211
- Abdullah Asif, H. M., Kamal, S., Rehman, A., Rasool, S., Hamid Akash, M. S. (2022). Synthesis, Characterization, and Enzyme Inhibition Properties of 1,2,4-Triazole Bearing Azinane Analogues. ACS Omega, 7 (36), 32360–32368. https://doi.org/10.1021/acsomega.2c03779
- Saeed, S., Shahzadi, I., Zahoor, A. F., Al-Mutairi, A. A., Kamal, S., Faisal, S. et al. (2024). Exploring theophylline-1,2,4-triazole tethered N-phenylacetamide derivatives as antimicrobial agents: unraveling mechanisms via structure-activity relationship, in vitro validation, and in silico insights. Frontiers in Chemistry, 12. https://doi.org/10.3389/fchem.2024.1372378
- Karpun, Y., Fedotov, S., Khilkovets, A., Karpenko, Y., Parchenko, V., Klochkova, Y. et al. (2023). An in silico investigation of 1,2,4-triazole derivatives as potential antioxidant agents using molecular docking, MD simulations, MM-PBSA free energy calculations and ADME predictions. Pharmacia, 70 (1), 139–153. https://doi.org/10.3897/pharmacia.70.e90783
- Gotsulya, А., Brytanova, T. (2022). Synthesis, properties and biological potential some condensed derivatives 1,2,4-triazole. Ankara Universitesi Eczacilik Fakultesi Dergisi, 46 (2), 308–321. https://doi.org/10.33483/jfpau.971602
- Ihnatova, T., Kaplaushenko, A., Frolova, Y., Pryhlo, E. (2021). Synthesis and antioxidant properties of some new 5-phenethyl-3-thio-1,2,4-triazoles. Pharmacia, 68 (1), 129–133. https://doi.org/10.3897/pharmacia.68.e53320
- Gitto, R., Vittorio, S., Bucolo, F., Peña-Díaz, S., Siracusa, R., Cuzzocrea, S. et al. (2022). Discovery of Neuroprotective Agents Based on a 5-(4-Pyridinyl)-1,2,4-triazole Scaffold. ACS Chemical Neuroscience, 13 (5), 581–586. https://doi.org/10.1021/acschemneuro.1c00849
- Saeed, S., Saif, M. J., Zahoor, A. F., Tabassum, H., Kamal, S., Faisal, S. et al. (2024). Discovery of novel 1,2,4-triazole tethered β-hydroxy sulfides as bacterial tyrosinase inhibitors: synthesis and biophysical evaluation through in vitro and in silico approaches. RSC Advances, 14 (22), 15419–15430. https://doi.org/10.1039/d4ra01252f
- Xu, X., Ding, M., Liu, K., Lv, F., Miao, Y., Liu, Y. et al. (2023). The synthesis and highly effective antibacterial properties of Cu-3, 5-dimethy l-1, 2, 4-triazole metal organic frameworks. Frontiers in Chemistry, 11. https://doi.org/10.3389/fchem.2023.1124303
- Khan, I., Rehman, W., Rahim, F., Hussain, R., Khan, S., Rasheed, L. et al. (2023). Synthesis and In Vitro α-Amylase and α-Glucosidase Dual Inhibitory Activities of 1,2,4-Triazole-Bearingbis-Hydrazone Derivatives and Their Molecular Docking Study. ACS Omega, 8 (25), 22508–22522. https://doi.org/10.1021/acsomega.3c00702
- Šermukšnytė, A., Kantminienė, K., Jonuškienė, I., Tumosienė, I., Petrikaitė, V. (2022). The Effect of 1,2,4-Triazole-3-thiol Derivatives Bearing Hydrazone Moiety on Cancer Cell Migration and Growth of Melanoma, Breast, and Pancreatic Cancer Spheroids. Pharmaceuticals, 15 (8), 1026. https://doi.org/10.3390/ph15081026
- Alam, M. M. (2024). New 1,2,4-triazole based eugenol derivatives as antiCOX-2 and anticancer agents. Journal of Umm Al-Qura University for Applied Sciences, 10 (3), 555–566. https://doi.org/10.1007/s43994-024-00127-z
- Gotsulya, A., Zazharskyi, V., Davydenko, P., Kulishenko, O., Parchenko, V., Brytanova, T. (2022). N’-(2-(5-((Theophylline-7-yl)methyl)-4-ethyl-1,2,4-triazole-3-ylthio)acetyl)isonicotinohydrazide As Antitubercular Agents. Hacettepe University Journal of the Faculty of Pharmacy, 42 (3), 149–155. https://doi.org/10.52794/hujpharm.1011368
- Mchichia, L., Belhassana, A., Aouidatea, A., Ghaleba, A., Lakhlifia, T., Bouachrinea, M. (2020). QSAR study of new compounds based on 1,2,4-triazole as potential anticancer agents. Physical chemistry research, 8 (1), 125–137. https://doi.org/10.22036/pcr.2019.204753.1685
- Fedotov, S., Goculya, A., Zaika, Y., Brytanova, T. (2023). Synthesis and properties of a series of 3-alkylthio derivatives 9-methylpyrazolo[1,5-d][1,2,4]triazolo[3,4-f][1,2,4]triazine. Ankara Universitesi Eczacilik Fakultesi Dergisi, 47 (2), 336–348. https://doi.org/10.33483/jfpau.1180794
- Fedotov, S. O., Hotsulia, A. S. (2023). Synthesis and properties of 6-(2,6-dichlorophenyl)-3- (3-methyl-1H-pyrazol-5-yl)-6,7-dihydro-5H-[1,2,4]triazolo[3,4-b] [1,3,4]thiadiazine-7-carboxylic acid and its salts. Current Issues in Pharmacy and Medicine: Science and Practice, 16 (2), 121–129. https://doi.org/10.14739/2409-2932.2023.2.279460
- Kumari, M., Tahlan, S., Narasimhan, B., Ramasamy, K., Lim, S. M., Shah, S. A. A. et al. (2021). Synthesis and biological evaluation of heterocyclic 1,2,4-triazole scaffolds as promising pharmacological agents. BMC Chemistry, 15 (1). https://doi.org/10.1186/s13065-020-00717-y
- Nashaan, F. A., Al-rawi, M. S. S. (2023). Design, synthesis, and biological activity of new thiazolidine-4-one derived from symmetrical 4-amino-1,2,4-triazole. Chemical Methodologies, 7, 187–196. https://doi.org/10.22034/chemm.2023.362512.1610
- Alsaad, H., Kubba, A., Tahtamouni, L. H., Hamzah, A. H. (2022). Synthesis, docking study, and structure activity relationship of novel anti-tumor 1, 2, 4 triazole derivatives incorporating 2-(2, 3- dimethyl aminobenzoic acid) moiety. Pharmacia, 69 (2), 415–428. https://doi.org/10.3897/pharmacia.69.e83158
- Azim, T., Wasim, M., Akhtar, M. S., Akram, I. (2021). An in vivo evaluation of anti-inflammatory, analgesic and anti-pyretic activities of newly synthesized 1, 2, 4 Triazole derivatives. BMC Complementary Medicine and Therapies, 21 (1). https://doi.org/10.1186/s12906-021-03485-x
- Abdulghani, S. M., Al-Rawi, M. S., Tomma, J. H. (2022). Synthesis of new 1,2,4-triazole derivatives with expected biological activities. Chemical Methodologies, 6, 59–66. https://doi.org/10.22034/chemm.2022.1.6
- Mikhina, E. A., Stepanycheva, D. V., Maksimova, V. P., Sineva, O. N., Markelova, N. N., Grebenkina, L. E. et al. (2024). Synthesis of Alkyl/Aryloxymethyl Derivatives of 1,2,4-Triazole-3-Carboxamides and Their Biological Activities. Molecules, 29 (20), 4808. https://doi.org/10.3390/molecules29204808
- Xia, M.-Y., Cai, Y.-X., Chen, J.-X., Zhao, X., Dong, H.-M., Yang, Z.-C. (2023). Synthesis, antimycobacterial evaluation, and molecular docking study of 1,2,4-triazole derivatives. Journal of Enzyme Inhibition and Medicinal Chemistry, 38 (1). https://doi.org/10.1080/14756366.2023.2229070
- Liu, X., Luo, J., Chen, J., Huang, P., He, G., Ye, X., Su, R., Lao, Y., Wang, Y., He, X., Zhang, J. (2024). The Neuroprotection of 1,2,4‐Triazole Derivative by Inhibiting Inflammation and Protecting BBB Integrity in Acute Ischemic Stroke. CNS Neuroscience & Therapeutics, 30 (11). https://doi.org/10.1111/cns.70113
- Radha, V. P., Prabakaran, M. (2022). Novel thiadiazole‐derived Schiff base ligand and its transition metal complexes: Thermal behaviour, theoretical study, chemo‐sensor, antimicrobial, antidiabetic and anticancer activity. Applied Organometallic Chemistry, 36 (11). https://doi.org/10.1002/aoc.6872
- Patil, S. K., Vibhute, B. T. (2021). Synthesis, characterization, anticancer and DNA photocleavage study of novel quinoline Schiff base and its metal complexes. Arabian Journal of Chemistry, 14 (8), 103285. https://doi.org/10.1016/j.arabjc.2021.103285
- Nafie, M. S., Boraei, A. T. A. (2022). Exploration of novel VEGFR2 tyrosine kinase inhibitors via design and synthesis of new alkylated indolyl-triazole Schiff bases for targeting breast cancer. Bioorganic Chemistry, 122, 105708. https://doi.org/10.1016/j.bioorg.2022.105708
- Bai, X., Ye, C., Liu, Z., Zhou, Z., Zhang, T. (2024). Synthesis and Biological Evaluation of Isoaurone Derivatives as Anti‐inflammatory Agents. Chemistry & Biodiversity, 22 (3). https://doi.org/10.1002/cbdv.202402073
- Abo-Elmagd, M. I., Hassan, R. M., Aboutabl, M. E., Amin, K. M., El-Azzouny, A. A., Aboul-Enein, M. N. (2024). Design, synthesis and anti-inflammatory assessment of certain substituted 1,2,4-triazoles bearing tetrahydroisoquinoline scaffold as COX 1/2-inhibitors. Bioorganic Chemistry, 150, 107577. https://doi.org/10.1016/j.bioorg.2024.107577
- Paprocka, R., Wiese-Szadkowska, M., Kołodziej, P., Kutkowska, J., Balcerowska, S., Bogucka-Kocka, A. (2023). Evaluation of Biological Activity of New 1,2,4-Triazole Derivatives Containing Propionic Acid Moiety. Molecules, 28 (9), 3808. https://doi.org/10.3390/molecules28093808
- Karnaš, M., Rastija, V., Vrandečić, K., Čosić, J., Kanižai Šarić, G., Agić, D. et al. (2024). Synthesis, antifungal, antibacterial activity, and computational evaluations of some novel coumarin-1,2,4-triazole hybrid compounds. Journal of Taibah University for Science, 18 (1). https://doi.org/10.1080/16583655.2024.2331456
- Hoffer, M. A. (2024). Exploring the reactivity of metal-ligand cooperative complexes with dioxazolones, terminal alkynes and 2-ethynylbenzyl alcohol. [Doctoral dissertation; The University of Western Ontario]. Available at: https://ir.lib.uwo.ca/etd/9913/
- Alshaye, N. A., Ibrahim, M. A. (2024). First approaches for the novel pyrido[1″,2″:2′,3′][1,2,4]triazolo[5′,1′:2,3][1,3]thiazolo[4,5-b]pyridines: synthesis, characterization and antimicrobial efficiency. Polycyclic Aromatic Compounds, 44 (9), 5899–5913. https://doi.org/10.1080/10406638.2023.2270555
- Mohamed, S. K., Anouar, E. H., Ahmad, S., Abbady, M. S., Abdel-Wadood, F. K., Qahtan, M. Q. M. et al. (2023). Synthesis, X-ray crystal structure, Hirshfeld surface analysis and computational investigation into the potential inhibitory action of novel 6-(p-tolyl)-2-((p-tolyl)thio)methyl-7H-[1,2,4]triazolo[5,1-b][1,3,4]thiadiazine inhibits the main protease of COVID-19. Journal of Biomolecular Structure and Dynamics, 41 (23), 14275–14284. https://doi.org/10.1080/07391102.2023.2180432
- Tati, A., Tabatabaee, M., Ardakani, A. A., Neumuller, B. (2023). In vitro evaluation, molecular docking, and Lipinski’s rule analysis of a new triazinone-based Schiff base for potential pharmacological applications. Progress in Ion Beam and Materials, 12 (3). https://doi.org/10.57647/pibm.2023.122316
- Mozharovskaia, P. N., Ivoilova, A. V., Malakhova, N. A., Drokin, R. A., Balin, I. A., Kozitsina, A. N. et al. (2024). Voltammetric Determination of a Potential Antiviral Drug Sodium Salt of 3-Nitro-4-Hydroxy-7-Methylthio-4H-[1,2,4]Triazolo[5,1-c][1,2,4]Triazinide Monohydrate. Journal of Analytical Chemistry, 79 (6), 733–739. https://doi.org/10.1134/s1061934824700114
- Salem, M. E., Mahrous, E. M., Ragab, E. A., Nafie, M. S., Dawood, K. M. (2023). Synthesis and Anti-Breast Cancer Potency of Mono- and Bis-(pyrazolyl[1,2,4]triazolo[3,4-b][1,3,4]thiadiazine) Derivatives as EGFR/CDK-2 Target Inhibitors. ACS Omega, 8 (38), 35359–35369. https://doi.org/10.1021/acsomega.3c05309
- Labunska, O.-L. I., Hunchak, V. M., Gutyj, B. V., Soltys, M. P. (2024). Study of acute and subacute toxicity of an immunostimulating drug based on the S-derivative of 1,2,4-triazole. Scientific Messenger of LNU of Veterinary Medicine and Biotechnologies, 26 (115), 145–152. https://doi.org/10.32718/nvlvet11521
- Kumar, S., Singh, P., Katiyar, R., Datusalia, A. K., Khatik, G. L. (2026). Chiral pool approach in design, synthesis, characterization, and evaluation of novel levorotatory- 1, 2, 4-triazole peptide-like AChE inhibitors. Journal of Molecular Structure, 1353, 144767. https://doi.org/10.1016/j.molstruc.2025.144767
- El Addali, A., El-Kiri, Z., El Boukili, A., Saufi, H., Boudad, L. (2024). Study of the protonation of 1, 2, 4-triazole. DFT Calculations. Journal of Chemistry and Technologies, 32 (4), 678–684. https://doi.org/10.15421/jchemtech.v32i4.300540
- Basha, N. J. (2023). Small Molecules as Anti‐inflammatory Agents: Molecular Mechanisms and Heterocycles as Inhibitors of Signaling Pathways. ChemistrySelect, 8 (9). https://doi.org/10.1002/slct.202204723
- Mehra, A., Mittal, A., Sangwan, R. (2025). Innovative Nitrogen-Based Heterocycles: Pioneering Advances in Anticancer Therapeutics. Current Chemical Biology, 19 (2), 92–140. https://doi.org/10.2174/0122127968335169250107180240
- Türker, L. (2023). Tautomers of 2,4-dihydro-3H-1,2,4-triazol-3-one and their Composites with NTO – A DFT Treatment. Earthline Journal of Chemical Sciences, 11 (1), 121–140. https://doi.org/10.34198/ejcs.11124.121140
- Biswas, S. (2022). Synthesis of some novel heterocyclic compounds composed of thiadiazole, azo and phenolic moieties and study of their antimicrobial property [Master's thesis; Bangladesh University of Engineering and Technology]. Available at: http://lib.buet.ac.bd:8080/xmlui/handle/123456789/6515
##submission.downloads##
Опубліковано
Як цитувати
Номер
Розділ
Ліцензія
Авторське право (c) 2026 Dmytro Dovbnia, Andrii Kaplaushenko, Oleksandr Panasenko, Mariia Panasenko, Volodymyr Salionov, Tetiana Ihnatova, Kaloyan Georgiev, Iliya Slavov

Ця робота ліцензується відповідно до Creative Commons Attribution 4.0 International License.
Наше видання використовує положення про авторські права Creative Commons Attribution 4.0 International License для журналів відкритого доступу.
Автори, які публікуються у цьому журналі, погоджуються з наступними умовами:
1. Автори залишають за собою право на авторство своєї роботи та передають журналу право першої публікації цієї роботи на умовах ліцензії Creative Commons Attribution 4.0 International License, котра дозволяє іншим особам вільно розповсюджувати опубліковану роботу з обов'язковим посиланням на авторів оригінальної роботи та першу публікацію роботи у цьому журналі.
2. Автори мають право укладати самостійні додаткові угоди щодо неексклюзивного розповсюдження роботи у тому вигляді, в якому вона була опублікована цим журналом (наприклад, розміщувати роботу в електронному сховищі установи або публікувати у складі монографії), за умови збереження посилання на першу публікацію роботи у цьому журналі.
3. Автори мають право зберігати остаточну прийняту версію статті в інституційному, тематичному або будь-якому іншому репозитарії з метою забезпечення видимості та доступності.







