Determining the optimal composition of low-basicity slags using pegmatite for electromelting processes
DOI:
https://doi.org/10.15587/1729-4061.2026.353249Keywords:
pegmatite, electric arc steel remelting, rheological properties of slag, refining of the meltingAbstract
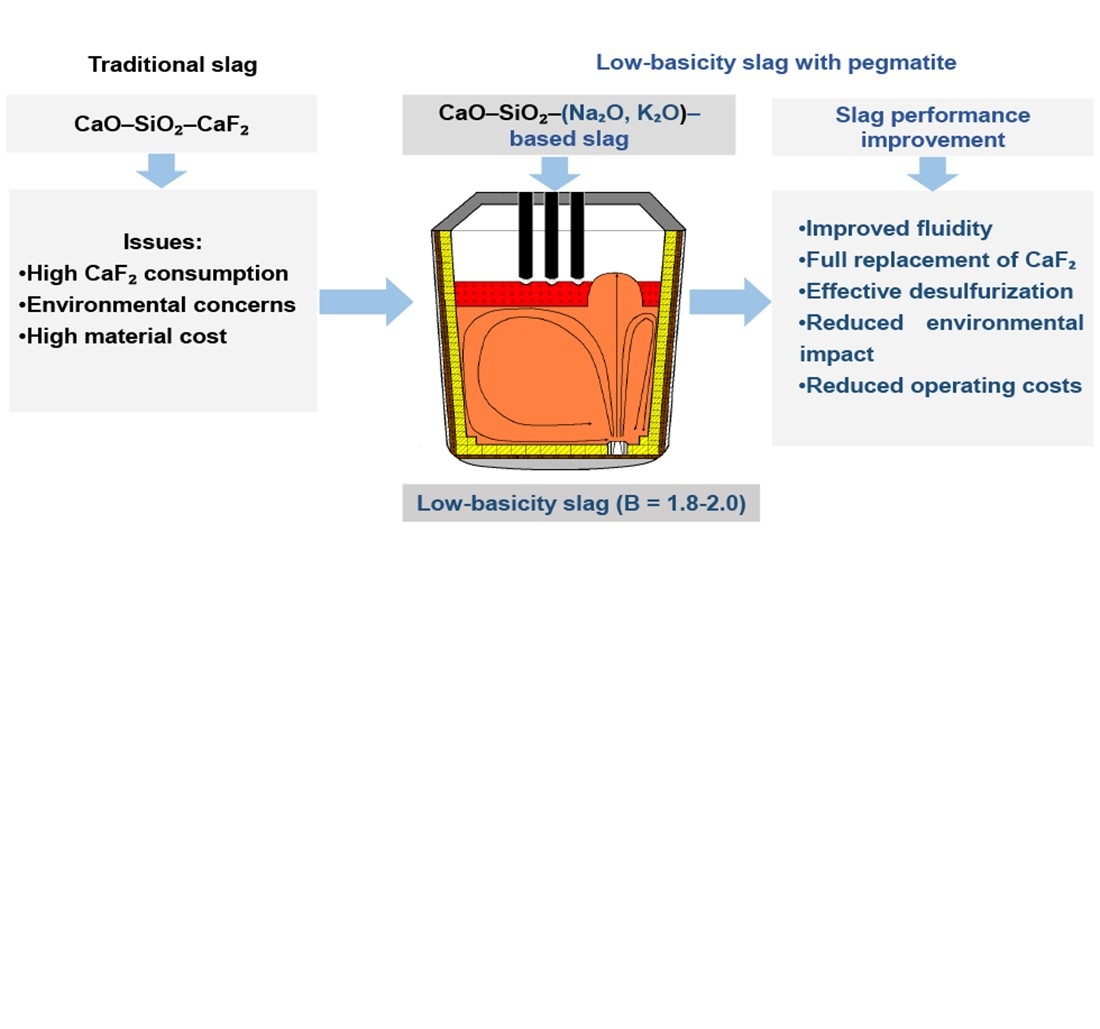
This study focuses on the process of slag formation and its performance during steelmaking using carbon charge, when remelting alloyed scrap in electric steelmaking units, and in secondary metallurgy units. One of the relevant issues is the use of alternative materials, such as alkali aluminosilicates, capable of replacing conventional slag components without compromising the quality of the slag.
This study reports scientifically proven conditions for replacing fluorspar in the composition of slags used in remelting technologies and secondary metallurgy with domestic mineral raw materials – pegmatites, which contain up to 10–15% of the total alkali metal oxides Na2O and K2O.
The effect of Na2O and K2O on the rheological characteristics of the slag melt in the CaO-SiO2+(Na2O, K2O) system has been confirmed. A series of experimental meltings was carried out to establish the slag-forming regime in a ladle-furnace when fluorspar in the solid slag-forming mixture is completely replaced with pegmatites.
A comparative analysis of the compositions of refining slags for the current industrial technology and the experimental technology has been performed. A significant increase in slag fluidity and a desulfurization level of the metal comparable to that of the current technology were established, despite a decrease in slag basicity to 1,8–2,0, which is consistent with the requirements of remelting technologies. Chemical analysis of the metal and slag compositions was conducted for the experimental meltings at the stages of semi-product melting and steel treatment in the ladle furnace. The metal from the experimental meltings fully met the requirements of the normative and technical documentation.
Thus, this work provides a theoretical justification for an innovative secondary-metallurgy technology using alkali aluminosilicate pegmatite as a part of slag forming mixtures. The metal quality indicators in terms of sulfur content confirm the effectiveness of the devised technology with partial or complete replacement of fluorspar with pegmatite during remelting or secondary steelmaking
References
- Li, S., Kong, L., Xu, Z. (2023). Effect of refining slag compositions on its melting property and desulphurization. High Temperature Materials and Processes, 42 (1). https://doi.org/10.1515/htmp-2022-0293
- Li, T., Li, G., Zhang, Z., Liu, Y., Wang, X. (2022). Fluoride vaporization and crystallization of CaF2–CaO–Al2O3–(La2O3) slag for vacuum electroslag remelting. Vacuum, 196, 110807. https://doi.org/10.1016/j.vacuum.2021.110807
- Peng, L., Jiang, Z., Geng, X., Liu, F., Li, H. (2019). Effect of B2O3 on the Crystallization Behavior of CaF2-Based Slag for Electroslag Remelting 9CrMoCoB Steel. Metals, 9 (12), 1331. https://doi.org/10.3390/met9121331
- An, B., Gu, Y., Ju, J., He, K. (2023). Fluoride Evaporation of Low-Fluoride CaF2-CaO-Al2O3-MgO-TiO2-(Na2O-K2O) Slag for Electroslag Remelting. Materials, 16 (7), 2777. https://doi.org/10.3390/ma16072777
- Huang, J., Zhang, L., Yu, W., Chen, J., Le, C., Ren, S. (2024). Extraction of Rare Earth and CaF2 from Rare Earth Calcium Thermal Reduction Slag by Using CaO Roasting–Acid Leaching Method. Minerals, 14 (10), 1001. https://doi.org/10.3390/min14101001
- Belashchenko, D. K., Ostrovski, O. I., Skvortsov, L. V. (2001). Molecular dynamics simulation of binary CaO–FeO, MgO–SiO2, FeO–SiO2, CaO–SiO2 and ternary CaO–FeO–SiO2 systems. Thermochimica Acta, 372 (1-2), 153–163. https://doi.org/10.1016/s0040-6031(01)00451-8
- Zhao, Q., Mei, X., Gao, L., Zhang, J., Wang, Z., Sun, L. et al. (2021). Fundamental Research on Fluorine-Free Ladle Furnace Slag for Axle Steel of Electric Multiple Unit Vehicles. Metals, 11 (12), 1973. https://doi.org/10.3390/met11121973
- Proidak, Yu. S., Gorobets, A. P., Zhadanos, O. V., Kamkina, L. V., Yaroshenko, Ya. O. (2025). Physical and chemical audits and comparative analyses of scrap remelting technology indicators for high-alloyed steel with special purposes using the duplex-slag process and the resource-saving mono-slag process. Theory and Practice of Metallurgy, 1, 67–72. https://doi.org/10.15802/tpm.1.2025.09
- Jancíková, Z., Švec, P. (2008). Prediction of chemical composition of refining slag with exploitation of artificial neural networks. Cybernetic letters: informatics, cybernetics and robotics, 2. Available at: http://www.cybletter.cz/index.php?id=67
- Tretiakov, Yu. I., Martyniuk, V. I., Subotin, A. V. et al. (2007). Mineralni resursy Ukrainy ta svitu. Kyiv, 560.
- Claquesin, J., Gibilaro, M., Massot, L., Lemoine, O., Bourges, G., Chamelot, P. (2021). Thermodynamic Properties of CaCl2-CaF2-CaO System: Phase Diagram Investigation. Materials Sciences and Applications, 12 (04), 139–151. https://doi.org/10.4236/msa.2021.124009
- Allibert, M. (1005). Slag Atlas. Verlag Stahleisen, 616.
- Chuiko, N. M., Rutkovskyi, V. B. et al. (1978). Nova tekhnolohiya vyplavky sharykopidshypnykovoi stali ShKh15 pid bilym shlakom. Visnyk vuziv. Chorna metalurhiya, 1, 38–43.
- Cho, M. K., Cheng, J., Park, J. H., Min, D. J. (2010). Hot Metal Desulfurization by CaO–SiO2–CaF2–Na2O Slag Saturated with MgO. ISIJ International, 50 (2), 215–221. https://doi.org/10.2355/isijinternational.50.215
- Berezhnyi, A. S. (1970). Bahatokomponentni systemy oksydiv. Kyiv, 544.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Yurii Proidak, Anton Gorobets, Oleksandr Zhadanos, Mariia Rybalchenko

This work is licensed under a Creative Commons Attribution 4.0 International License.
The consolidation and conditions for the transfer of copyright (identification of authorship) is carried out in the License Agreement. In particular, the authors reserve the right to the authorship of their manuscript and transfer the first publication of this work to the journal under the terms of the Creative Commons CC BY license. At the same time, they have the right to conclude on their own additional agreements concerning the non-exclusive distribution of the work in the form in which it was published by this journal, but provided that the link to the first publication of the article in this journal is preserved.
A license agreement is a document in which the author warrants that he/she owns all copyright for the work (manuscript, article, etc.).
The authors, signing the License Agreement with TECHNOLOGY CENTER PC, have all rights to the further use of their work, provided that they link to our edition in which the work was published.
According to the terms of the License Agreement, the Publisher TECHNOLOGY CENTER PC does not take away your copyrights and receives permission from the authors to use and dissemination of the publication through the world's scientific resources (own electronic resources, scientometric databases, repositories, libraries, etc.).
In the absence of a signed License Agreement or in the absence of this agreement of identifiers allowing to identify the identity of the author, the editors have no right to work with the manuscript.
It is important to remember that there is another type of agreement between authors and publishers – when copyright is transferred from the authors to the publisher. In this case, the authors lose ownership of their work and may not use it in any way.





