Determination of the formation mechanism of zinc and nickel layered double hydroxides
DOI:
https://doi.org/10.15587/1729-4061.2026.358469Keywords:
potentiometric titration, formation mechanism, Zn-Al LDH, Ni-Al LDH, Ni(II)-Ni(III) LDHAbstract
The object of the study – the formation mechanism of Zn-Al nitrate, Zn-Al tripolyphosphate, Ni-Al carbonate and Ni(II)-Ni(III) carbonate layered-double hydroxides (LDHs).
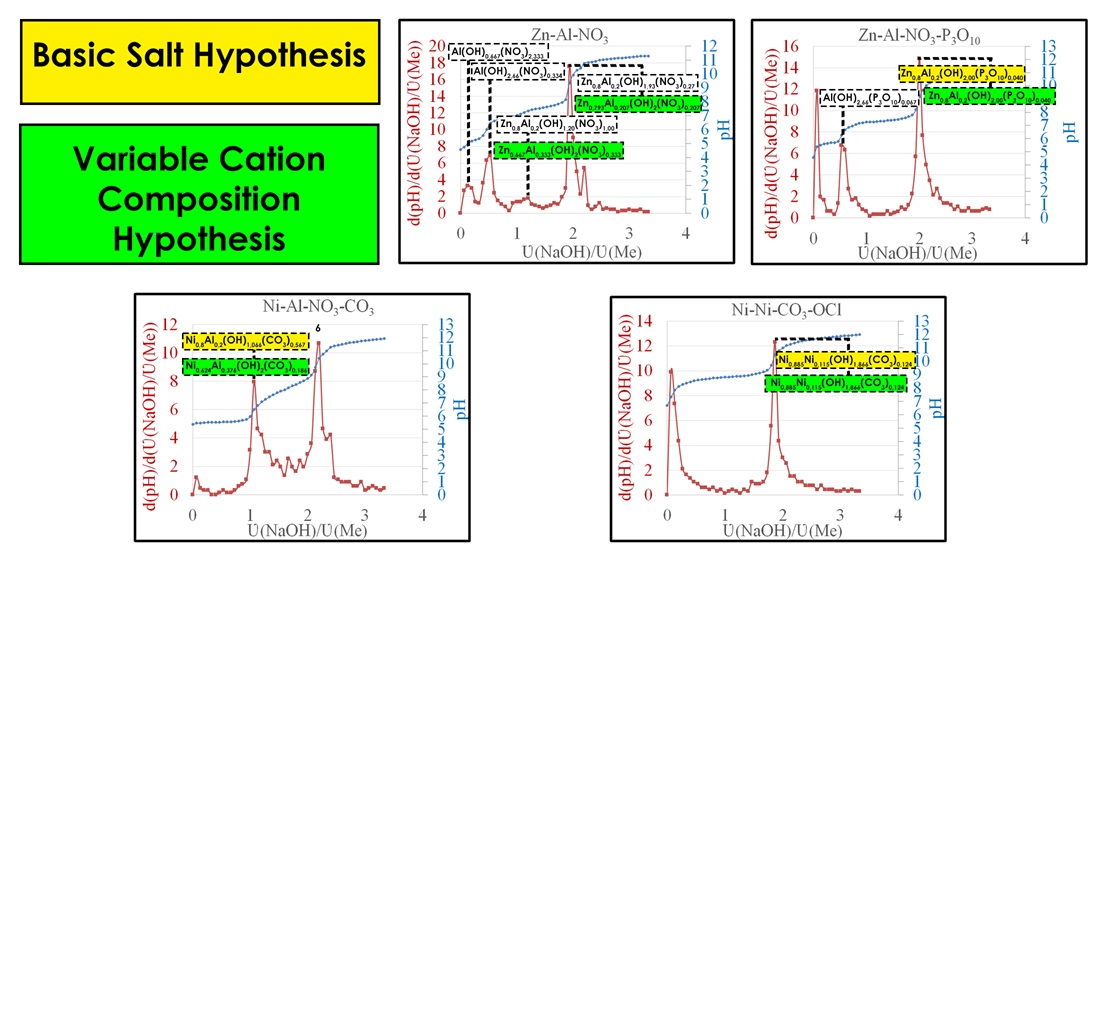
To calculate the composition of the precipitate at the LDH formation stage, the following hypotheses were proposed:
1) basic salt hypothesis (BSH): all cations completely precipitate, then OH- ions replace the anions;
2) variable cation composition hypothesis (VCCH): the precipitate is formed by a saturated "guest" cation, and subsequently the amount of the "host" cation increases.
Potentiometric titration with a glass electrode was used to determine the LDH formation mechanism.
A two-stage formation mechanism was observed for Zn-Al-NO3 LDH. At the first stage, aluminum hydroxide forms (pH = 6.47) via the formation of the hydroxo salt Al(OH)0,667(NO3)2,333 (pH = 4.98). At the second stage, LDH formation proceeds through intermediate stages: at pH = 7.36, the is composition Zn0,8Al0,2(OH)1,20(NO3)1,00 (BSH) or Zn0,667Al0,333(OH)2(NO3)1,667 (VCCH), at pH = 9.35, the composition is Zn0,8Al0,2(OH)1,93(NO3)0,27 (BSH) or Zn0,793Al0,207(OH)2(NO3)0,204 (VCCH).
A two-stage mechanism is characteristic of Zn-Al-P3O10 LDH. At pH = 6.25, the hydroxo salt Al(OH)2,66(P3O10)0,067 is formed, whereas at pH = 9.25, LDH with the composition Zn0,8Al0,2(OH)2,00(P3O10)0,040 is obtained. For Zn-Al LDH, a pH range of 7–10 is recommended.
It was found that during the formation of Ni-Al-CO3 LDH, the stage of Al hydroxocompound formation is absent due to the complete irreversible hydrolysis of Al3+ in the presence of At pH = 6.5, the precipitate Ni0,8Al0,2(OH)1,066(CO3)0,567 (BSH) or Ni0,624Al0,376(OH)2(CO3)0,376 (VCCH) is formed, which is transformed into LDH upon further alkalization. The synthesis of Ni-Al-CO3 LDH should be carried out at pH = 8–11.
A single-stage formation mechanism is determined for Ni(II)-Ni(III)-CO3 LDH, with a recommended synthesis pH range of 10–11.
Comparative analysis shows that the VCCH better describes the change in the precipitate composition during LDH formation
References
- Nalawade, P., Aware, B., Kadam, V., Hirlekar, R. (2009). Layered double hydroxides: A review. Journal of Scientific and Industrial Research, 68, 267–272. Available at: https://www.hazemsakeek.net/wp-content/uploads/2021/06/LDH.pdf
- Kovalenko, V., Kotok, V., Murashevych, B. (2023). Layered Double Hydroxides as the Unique Product of Target Ionic Construction for Energy, Chemical, Foods, Cosmetics, Medicine and Ecology Applications. The Chemical Record, 24 (2). https://doi.org/10.1002/tcr.202300260
- Kovalenko, V., Borysenko, A., Kotok, V., Nafeev, R., Verbitskiy, V., Melnyk, O. (2022). Determination of the dependence of the structure of Zn-Al layered double hydroxides, as a matrix for functional anions intercalation, on synthesis conditions. Eastern-European Journal of Enterprise Technologies, 1 (12 (115)), 12–20. https://doi.org/10.15587/1729-4061.2022.252738
- Sasai, R., Sato, H., Sugata, M., Fujimura, T., Ishihara, S., Deguchi, K. et al. (2019). Why Do Carbonate Anions Have Extremely High Stability in the Interlayer Space of Layered Double Hydroxides? Case Study of Layered Double Hydroxide Consisting of Mg and Al (Mg/Al = 2). Inorganic Chemistry, 58 (16), 10928–10935. https://doi.org/10.1021/acs.inorgchem.9b01365
- Wijitwongwan, R. (Ploy), Intasa-ard, S. (Grace), Ogawa, M. (2019). Preparation of Layered Double Hydroxides toward Precisely Designed Hierarchical Organization. ChemEngineering, 3 (3), 68. https://doi.org/10.3390/chemengineering3030068
- Khan, A. I., Ragavan, A., Fong, B., Markland, C., O’Brien, M., Dunbar, T. G. et al. (2009). Recent Developments in the Use of Layered Double Hydroxides as Host Materials for the Storage and Triggered Release of Functional Anions. Industrial & Engineering Chemistry Research, 48 (23), 10196–10205. https://doi.org/10.1021/ie9012612
- Izaddin Sheikh Mohd Ghazali, S. A., Dzulkifli, N. N., Abdullah, A., Fatimah, I., Biswas, H. S., Kovalenko, V., Poddar, S. (2025). Synthesis of novel ternary herbicide-layered double hydroxide hybrids via the ion exchange method. Pure and Applied Chemistry, 97 (5), 451–459. https://doi.org/10.1515/pac-2024-0336
- Mohd Shah, A. H., Izaddin Sheikh Ghazali, S. A., Md Norizan, N. E., Roslan, N. J., Dzulkifli, N. N., Zaki, H. M. et al. (2025). Design and evaluation of zinc-aluminium layered double hydroxide-palmitic acid nanocomposites: synthesis, characterization and antimicrobial properties. Malaysian Journal of Analytical Sciences, 29 (3), 1304. Available at: https://mjas.analis.com.my/mjas/v29_n3/pdf/Mohd%20Shah_29_3_1304.pdf
- Burmistr, M. V., Boiko, V. S., Lipko, E. O., Gerasimenko, K. O., Gomza, Yu. P., Vesnin, R. L. et al. (2014). Antifriction and Construction Materials Based on Modified Phenol-Formaldehyde Resins Reinforced with Mineral and Synthetic Fibrous Fillers. Mechanics of Composite Materials, 50 (2), 213–222. https://doi.org/10.1007/s11029-014-9408-0
- Mohd Shah, A. H., Ahmad, A., Dzulkifli, N. N., Mohd Zaki, H., Fatimah, I., Kovalenko, V., Sheikh Mohd Ghazali, S. A. I. (2026). pH-responsive LDH-palmitic acid nanohybrids for controlled drug delivery. Pure and Applied Chemistry. https://doi.org/10.1515/pac-2025-0602
- Kesavan Pillai, S., Kleyi, P., de Beer, M., Mudaly, P. (2020). Layered double hydroxides: An advanced encapsulation and delivery system for cosmetic ingredients-an overview. Applied Clay Science, 199, 105868. https://doi.org/10.1016/j.clay.2020.105868
- Kameliya, J., Verma, A., Dutta, P., Arora, C., Vyas, S., Varma, R. S. (2023). Layered Double Hydroxide Materials: A Review on Their Preparation, Characterization, and Applications. Inorganics, 11 (3), 121. https://doi.org/10.3390/inorganics11030121
- Zhang, G., Zhang, X., Meng, Y., Pan, G., Ni, Z., Xia, S. (2020). Layered double hydroxides-based photocatalysts and visible-light driven photodegradation of organic pollutants: A review. Chemical Engineering Journal, 392, 123684. https://doi.org/10.1016/j.cej.2019.123684
- Yousaf, M. H., Shahid, M., Ahmad, N., Ali, M., Yousaf, G. (2023). Catalytic Hydrogenation of Organic Polymers by Mixed Transition Metal Oxides. International Journal of Innovative Science and Research Technology, 8 (5), 3234–3241. https://doi.org/10.5281/zenodo.8049671
- Kotok, V., Kovalenko, V., Malyshev, V. (2017). Comparison of oxygen evolution parameters on different types of nickel hydroxide. Eastern-European Journal of Enterprise Technologies, 5 (12 (89)), 12–19. https://doi.org/10.15587/1729-4061.2017.109770
- Kovalenko, V., Kotok, V., Kovalenko, I. (2018). Activation of the nickel foam as a current collector for application in supercapacitors. Eastern-European Journal of Enterprise Technologies, 3 (12 (93)), 56–62. https://doi.org/10.15587/1729-4061.2018.133472
- Kotok, V. A., Kovalenko, V. L., Solovov, V. A., Kovalenko, P. V., Ananchenko, B. A. (2018). Effect of deposition time on properties of electrochromic nickel hydroxide films prepared by cathodic template synthesis. ARPN Journal of Engineering and Applied Sciences, 13 (9), 3076–3086. Available at: https://www.arpnjournals.org/jeas/research_papers/rp_2018/jeas_0518_7034.pdf
- Kotok, V. A., Kovalenko, V. L. (2019). Non-Metallic Films Electroplating on the Low-Conductivity Substrates: The Conscious Selection of Conditions Using Ni(OH)2Deposition as an Example. Journal of The Electrochemical Society, 166 (10), D395–D408. https://doi.org/10.1149/2.0561910jes
- Solovov, V. A., Nikolenko, N. V., Kovalenko, V. L., Kotok, V. A., Burkov, A. А., Kondrat’ev, D. A. et al. (2018). Synthesis of Ni(II)-Ti(IV) Layered Double Hydroxides Using Coprecipitation At High Supersaturation Method. ARPN Journal of Engineering and Applied Sciences, 13 (24), 9652–9656. Available at: https://www.arpnjournals.org/jeas/research_papers/rp_2018/jeas_1218_7500.pdf
- Alibakhshi, E., Ghasemi, E., Mahdavian, M., Ramezanzadeh, B. (2017). A comparative study on corrosion inhibitive effect of nitrate and phosphate intercalated Zn-Al- layered double hydroxides (LDHs) nanocontainers incorporated into a hybrid silane layer and their effect on cathodic delamination of epoxy topcoat. Corrosion Science, 115, 159–174. https://doi.org/10.1016/j.corsci.2016.12.001
- Roslan, N. J., Mohd Shah, A. H., Dzulkifli, N. N., Saharuddin, T. S. T., Rusmin, R., Jasni, A. H. et al. (2025). Interlayer Expansion and Morphological Transformation of Zinc-Aluminium Anionic Clay through Methyl Orange Intercalation. Malaysian Journal of Chemistry, 27 (3), 254–260. https://doi.org/10.55373/mjchem.v27i3.254
- Kovalenko, V., Kotok, V. (2020). Tartrazine-intercalated Zn–Al layered double hydroxide as a pigment for gel nail polish: synthesis and characterisation. Eastern-European Journal of Enterprise Technologies, 3 (12 (105)), 29–37. https://doi.org/10.15587/1729-4061.2020.205607
- Vazquez, R. N. M., Nuñez, C. P., Kovalenko, V., Kotok, V., Moisés, F. P. P., Lamas, A. M. M., Arízaga, G. G. C. (2023). Electron Transfer within an Antioxidant Powder Composite with Layered Double Hydroxide Nanoparticles and Tomato Extract. Biointerface Research in Applied Chemistry, 13 (3), 257. https://doi.org/10.33263/briac133.257
- Andrade, K. N., Pérez, A. M. P., Arízaga, G. G. C. (2019). Passive and active targeting strategies in hybrid layered double hydroxides nanoparticles for tumor bioimaging and therapy. Applied Clay Science, 181, 105214. https://doi.org/10.1016/j.clay.2019.105214
- Gao, S.-P., Lu, T.-H., Li, S.-P., Zhong, H. (2009). The mechanism on the pH value influencing the property of glutamic acid/layered double hydroxide compounds. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 351 (1-3), 26–29. https://doi.org/10.1016/j.colsurfa.2009.09.021
- Bukhtiyarova, M. V. (2019). A review on effect of synthesis conditions on the formation of layered double hydroxides. Journal of Solid State Chemistry, 269, 494–506. https://doi.org/10.1016/j.jssc.2018.10.018
- Seron, A., Delorme, F. (2008). Synthesis of layered double hydroxides (LDHs) with varying pH: A valuable contribution to the study of Mg/Al LDH formation mechanism. Journal of Physics and Chemistry of Solids, 69 (5-6), 1088–1090. https://doi.org/10.1016/j.jpcs.2007.10.054
- Grégoire, B., Ruby, C., Carteret, C. (2013). Hydrolysis of mixed Ni2+–Fe3+ and Mg2+–Fe3+ solutions and mechanism of formation of layered double hydroxides. Dalton Transactions, 42 (44), 15687. https://doi.org/10.1039/c3dt51521d
- Xu, Z. P., Braterman, P. S. (2014). Layered Double Hydroxides: Self-Assembly and Multiple Phases. Dekker Encyclopedia of Nanoscience and Nanotechnology, Third Edition, 2056–2066. https://doi.org/10.1081/e-enn3-120013852
- Boclair, J. W., Braterman, P. S. (1999). Layered Double Hydroxide Stability. 1. Relative Stabilities of Layered Double Hydroxides and Their Simple Counterparts. Chemistry of Materials, 11 (2), 298–302. https://doi.org/10.1021/cm980523u
- Kovalenko, V., Kotok, V. (2019). “Smart” anticorrosion pigment based on layered double hydroxide: construction and characterization. Eastern-European Journal of Enterprise Technologies, 4 (12 (100)), 23–30. https://doi.org/10.15587/1729-4061.2019.176690
- Kovalenko, V., Andreiev, D., Kotok, V., Baskevich, A., Medianyk, V., Sukhomlyn, D., Verbitskiy, V. (2026). Determination of the possibility of obtaining ballastless Ni(II)-Ni(III) layered double hydroxides as promising active substances for supercapacitors and electrocatalysis. Eastern-European Journal of Enterprise Technologies, 1 (6 (139)), 6–15. https://doi.org/10.15587/1729-4061.2026.352268
- Kovalenko, V., Kotok, V., Girenko, D., Nikolenko, M., Andreiev, D., Verbitskiy, V. et al. (2023). Investigation of the mechanism of nickel hydroxide formation from nickel nitrate. Eastern-European Journal of Enterprise Technologies, 1 (6 (121)), 58–65. https://doi.org/10.15587/1729-4061.2023.272673
- Johari, N. A., Mahathir, N. F. A., Shah, N. A., Dzulkifli, N. N., Fatimah, I., Adam, N. et al. (2023). Zinc Layered Hydroxide 2-methyl-4-chlorophenoxyacetate: Synthesis via ZnO, Characterization and Effect on Seed Germination. Trends in Sciences, 20 (12), 6943. https://doi.org/10.48048/tis.2023.6943
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Vadym Kovalenko, Anastasiia Borysenko, Dmytro Andreiev, Anton Dopira, Oleksii Demchenko, Valerii Kotok, Volodymyr Medianyk, Dmytro Sukhomlyn, German Shuklin, Volodymyr Verbitskiy

This work is licensed under a Creative Commons Attribution 4.0 International License.
The consolidation and conditions for the transfer of copyright (identification of authorship) is carried out in the License Agreement. In particular, the authors reserve the right to the authorship of their manuscript and transfer the first publication of this work to the journal under the terms of the Creative Commons CC BY license. At the same time, they have the right to conclude on their own additional agreements concerning the non-exclusive distribution of the work in the form in which it was published by this journal, but provided that the link to the first publication of the article in this journal is preserved.
A license agreement is a document in which the author warrants that he/she owns all copyright for the work (manuscript, article, etc.).
The authors, signing the License Agreement with TECHNOLOGY CENTER PC, have all rights to the further use of their work, provided that they link to our edition in which the work was published.
According to the terms of the License Agreement, the Publisher TECHNOLOGY CENTER PC does not take away your copyrights and receives permission from the authors to use and dissemination of the publication through the world's scientific resources (own electronic resources, scientometric databases, repositories, libraries, etc.).
In the absence of a signed License Agreement or in the absence of this agreement of identifiers allowing to identify the identity of the author, the editors have no right to work with the manuscript.
It is important to remember that there is another type of agreement between authors and publishers – when copyright is transferred from the authors to the publisher. In this case, the authors lose ownership of their work and may not use it in any way.








